FDA warns of cardiac risk with stress test agents

(HealthDay)—The use of cardiac nuclear stress test agents comes with a rare but serious risk of heart attack and death, according to a warning sent by the U.S. Food and Drug Administration to health care professionals.
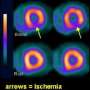
Lexiscan (regadenoson) and Adenoscan (adenosine), used in cardiac stress tests in patients who cannot exercise adequately, help identify coronary artery disease by dilating heart arteries and increasing blood flow. The risk stems from reduced blood flow to obstructed areas, which may result in potentially fatal heart attack.
The FDA has approved drug label changes for the two drugs to highlight these risks and update recommendations for their use. At this time, it is not clear if there is any difference in risk between Lexiscan and Adenoscan.
According to the agency, health care professionals should "screen all nuclear stress test candidates for their suitability to receive Lexiscan or Adenoscan. Avoid using these drugs in patients with signs or symptoms of unstable angina or cardiovascular instability, as these patients may be at greater risk for serious cardiovascular adverse reactions."
More information: More Information
Copyright © 2013 HealthDay. All rights reserved.