Early trial shows injectable agent illuminates cancer during surgery
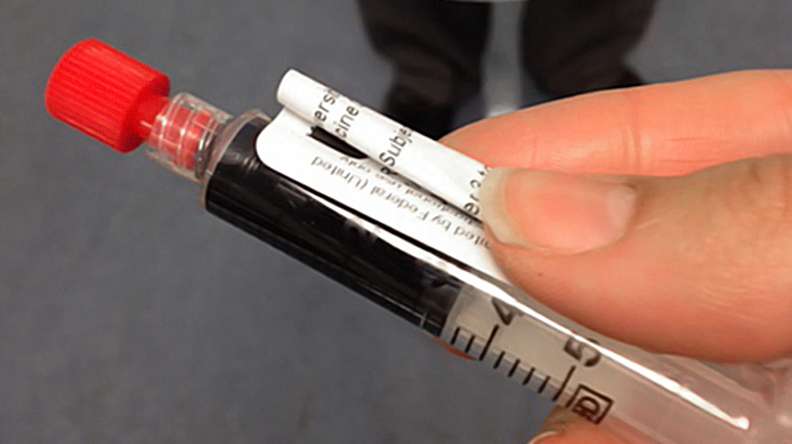
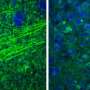
Doctors at the Duke University School of Medicine have tested a new injectable agent that causes cancer cells in a tumor to fluoresce, potentially increasing a surgeon's ability to locate and remove all of a cancerous tumor on the first attempt. The imaging technology was developed through collaboration with scientists at Duke, the Massachusetts Institute of Technology (MIT) and Lumicell Inc.
According to findings published January 6 in Science Translational Medicine, a trial at Duke University Medical Center in 15 patients undergoing surgery for soft-tissue sarcoma or breast cancer found that the injectable agent, a blue liquid called LUM015 (loom - fifteen), identified cancerous tissue in human patients without adverse effects.
Cancer surgeons currently rely on cross-sectional imaging such as MRIs and CT scans to guide them as they remove a tumor and its surrounding tissue. But in many cases some cancerous tissue around the tumor is undetected and remains in the patient, sometimes requiring a second surgery and radiation therapy.
"At the time of surgery, a pathologist can examine the tissue for cancer cells at the edge of the tumor using a microscope, but because of the size of cancer it's impossible to review the entire surface during surgery," said senior author David Kirsch, M.D., Ph.D., a professor of radiation oncology and pharmacology and cancer biology at Duke University School of Medicine. "The goal is to give surgeons a practical and quick technology that allows them to scan the tumor bed during surgery to look for any residual fluorescence."
Researchers around the globe are pursuing techniques to help surgeons better visualize cancer, some using a similar mechanism as LUM015, which is activated by enzymes. But the Duke trial described in the journal is the first protease-activated imaging agent for cancer that has been tested for safety in humans, Kirsch said.
LUM015 was developed by Lumicell, a company started by researchers at MIT and involving Kirsch. In companion experiments in mice described in the journal, LUM015 accumulated in tumors where it creates fluorescence in tumor tissue that is on average five times brighter than regular muscle. The resulting signals aren't visible to the naked eye and must be detected by a handheld imaging device with a sensitive camera, which Lumicell is also developing, Kirsch said.
In the operating room after a tumor is removed, surgeons would place the handheld imaging device on the cut surface. The device would alert them to areas with fluorescent cancer cells.
Going into surgery, the goal is always to remove 100 percent of the tumor, plus a margin of normal tissue around the edges, explained senior author Brian Brigman, M.D., Ph.D., chief of orthopedic oncology at Duke. Pathologists then analyze the margins over several days and determine whether they are clear.
"This pathologic technique to determine whether tumor remains in the patient is the best system we have currently, and has been in use for decades, but it's not as accurate as we would like," said Brigman, who is also the director of the sarcoma program at the Duke Cancer Institute. "If this technology is successful in subsequent trials, it would significantly change our treatment of sarcoma. If we can increase the cases where 100 percent of the tumor is removed, we could prevent subsequent operations and potentially cancer recurrence. Knowing where there is residual disease can also guide radiation therapy, or even reduce how much radiation a patient will receive."
Researchers at Massachusetts General Hospital are currently evaluating the safety and efficacy of LUM015 and the Lumicell imaging device in a prospective study of 50 women with breast cancer. Afterward, Kirsch said, multiple institutions would likely evaluate whether the technology can decrease the number of patients needing subsequent operations following initial breast cancer removal.
More information: "A mouse-human phase 1 co-clinical trial of a protease-activated fluorescent probe for imaging cancer," Science Translational Medicine, stm.sciencemag.org/lookup/doi/ … scitranslmed.aad0293