Nerve stimulation with the help of implantable mini solar cells
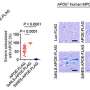
An international research team has successfully developed and tested a concept in which nerves are stimulated with light pulses. The method provides considerable advantages for medicine and opens up a wide range of possible ...
Apr 7, 2022
0
93