First Look at the 'Birth' of a Retina Cell
Scientists at the University of Michigan Kellogg Eye Center have gained new insight into the way an embryonic retina cell develops and then commits itself to a specific role. They have observed a small window of opportunity during which a cell has been designated to play a particular role, but has not yet begun to function as such.
The discovery provides a new vantage point for understanding how a healthy visual system develops. It also raises the possibility of re-directing the production of cell types as needed to stave off eye disease.
The study reports on the role of the gene regulator NRL, which was previously shown to be essential for the development of rods, the light-sensing cells required for vision.
Anand Swaroop, Ph.D., Harold F. Falls Collegiate Professor of Ophthalmology and Visual Sciences and Professor of Human Genetics, is senior author of both studies, the latest of which appeared in the March 7 issue of the Proceedings of the National Academy of Sciences (PNAS).
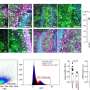
In PNAS, Swaroop's team reported that NRL is the earliest marker of rod precursors, or cells that are fated to become rods. They achieved this unique and early view of rod development by creating a mouse model using an NRL regulatory DNA sequence to produce a protein that appears as fluorescent green when exposed to blue light. This fluorescent protein allows scientists to identify even a few cells that are destined to be rods at very early stages of development.
“For the first time we were able to see retina cells during early development, allowing us to pinpoint the exact time at which rods are ‘born,’” says Swaroop. “Because the cells have been tagged, we can watch them at each step as they develop into mature and functional rods.” Kellogg scientists then purified the rods at various stages and generated the profile of genes at each stage. This, for the first time, provided a handle for investigating the precise process of rod differentiation.
Rods, along with cones, are photoreceptors, which account for 70 percent to 80 percent of all cells in the adult retina. Rods greatly outnumber cones in the mouse and in humans.
Damage to photoreceptors is at the root of eye diseases such as diabetic retinopathy, retinitis pigmentosa, and macular degeneration. In most instances, including age-related macular degeneration, rod photoreceptors die before cones.
As the mouse eye develops, rods start out as stem cells, meaning they have not yet been assigned a function and could theoretically grow to become any kind of cell. Along the way, these cells change their gene expression and acquire competence to become a specific cell type. At a certain point, cell division stops, and the cell is fated to perform a certain function.
“We can now view rod precursor cells at a crucial juncture,” says Swaroop. “They are committed to becoming rod cells, but they are still adaptable and have not yet ‘become' that type of cell.” In mouse models, it takes five to fourteen days for rod cells to become functional, whereas in humans this time period is four to five weeks during gestation.
The Swaroop research team also confirmed that when NRL is absent, a rod precursor will change its course and acquire the identity of a cone. “This finding in particular implies the existence of pools of progenitor cells with competence to become either a rod or a cone,” explains Swaroop. “We suggest that during early stages of development, these cells are not completely committed to a specific fate; there is an opportunity for regulators such as NRL to instruct these cells to produce rods or cones.”
With the ability to see greater detail in events along molecular pathways, Swaroop is enthusiastic about finding new methods for disrupting the disease process. There may also be opportunities to promote the production of new rods and cones when others die off.
In future studies, the Kellogg scientist plans to use this new mouse model in conjunction with other mouse models of specific diseases, such as macular and other retinal degenerations. Gene profiling of rods and cones in disease models at various stages of pathogenesis could help scientists identify molecular targets for drug treatment.
Swaroop is hopeful that his research group can use the fluorescent protein to illuminate the chain of events that occurs when a gene mutation interferes with rod — or even cone — development. “This model may give us the earliest look yet at some devastating diseases we are all eager to cure,” he says.
Citation: Targeting of GFP to newborn rods by Nrl promoter and temporal expression profiling of flow-sorted photoreceptors, PNAS, March 7, 2006; vol. 103, no. 10: 3890-3895
Source: University of Michigan