Researchers Make Vitamin E Offshoot A Potent Cancer Killer
Researchers here have learned how a derivative of vitamin E causes the death of cancer cells. The researchers then used that knowledge to make the agent an even more potent cancer killer.
The compound, called vitamin E succinate, or alpha tocopheryl succinate, is taken by some people as a nutritional supplement, mainly for its antioxidant properties. In addition, it has a weak ability to kill cancer cells, and it has been tested as a cancer chemopreventive agent.
The substance kills cancer cells by causing them to undergo a natural process known as programmed cell death, or apoptosis. Until now, no one knew how the agent caused this to happen.
These findings answer that question and also indicate that the molecule's antitumor activity is separate from its antioxidant effect.
The study, led by researchers with The Ohio State University Comprehensive Cancer Center – Arthur G. James Cancer Hospital and Richard J. Solove Research Institute (OSUCCC-James), is published in the April 28 issue of the Journal of Biological Chemistry.
“Our findings could lead to a potent chemopreventive agent that has both strong anticancer and antioxidant properties,” says principal investigator Ching-Shih Chen, professor of pharmacy and of internal medicine and a researcher with the OSUCCC-James.
“Such an agent might help reduce the risk of prostate, colon and other cancers.”
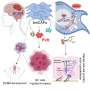
Chen and his collaborators found that vitamin E succinate works by blocking a protein called Bcl-xL. The protein, which is made by healthy cells, is often present at abnormally high levels in cancer cells and protects them from dying when they should.
Using computer modeling, the researchers found that the vitamin E derivative works because it lodges in a groove in the structure of the Bcl-xL protein, disabling it.
However, the vitamin E molecule has a long, coiled, protruding tail that keeps the molecule from fitting tightly, and more effectively, into the groove.
“Once we identified how the agent and the protein interact, we asked how we could improve that interaction,” Chen says.
The scientists found that a relatively simple process of altering the molecule's structure – basically cutting the tail short – allowed a tighter fit and improved the agent's ability to kill cancer cells by five- to ten-fold in laboratory tests.
“Overall, out findings are proof of the principle that this drug can kill cancer cells very effectively but does very little damage to healthy cells,” Chen says.
Source: Ohio State University