Analysis of Chinese AIDS epidemic shows surprising patterns
The mountainous Chinese province of Yunnan is tucked into the country’s southwest corner, a scenic region that borders Burma, Laos and Vietnam. The province shares its rugged topography with the surrounding countries, but it shares a less favorable trait as well: a growing AIDS epidemic, driven by a thriving underground commercial sex trade and by heroin smuggled in from the opium-growing regions of its three neighbors, an area known as the “golden triangle.”
Yunnan was the point of entry for AIDS in China, with the first cases appearing in 1989, and the mutations of the virus that exist there are far more diverse than anywhere else in the country.
Researchers at Rockefeller University and the Aaron Diamond AIDS Research Center (ADARC) have been doing work in Yunnan province for close to a decade. Because all the forms of HIV in China can be traced back to the area, they’re interested in how different subtypes of the virus are related to the epidemic’s spread and how they’re evolving. Their most recent findings, announced last fall, suggest that the virus may be spread via sexual transmission more quickly than anyone believed, resulting in an HIV subtype that has the potential to kick off a whole new Chinese epidemic.
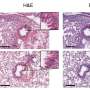
The new research, led by assistant professor Zheiwei Chen and published in a recent issue of PLoS Medicine, characterizes the different strains of HIV that are currently in circulation in Yunnan and, for the first time, associates different subtypes with different groups of people. “We wanted to do a basic molecular epidemiology study covering the entire province,” Chen says. So his research group — which included colleagues from the Yunnan Center for Disease Control and Prevention in Kunming, China, and David Ho, Rockefeller's Irene Diamond Professor and ADARC’s scientific director — gathered blood samples from 321 HIV-infected people. The subjects lived throughout the province and represented a wide cross-section of multiple communities, ethnicities and professions.
Because the virus evolves so quickly, the researchers could track HIV subtypes using their genetic variation and were able to determine that certain subtypes were associated with certain populations. Of those people who had known risk factors, intravenous drug users composed nearly 91 percent of the virus’s “BC” variants, while 85 percent of the “AE” variants appeared to be sexually transmitted. “We thought that the majority of HIV cases in Yunnan were intravenous drug users,” Chen says. But by linking particular risk factors to genetic subtypes, they could see a new pattern emerging. “A substantial number of people acquired their infections through sexual contact.”
Chen notes that the AE variants are also sexually transmitted strains in Thailand, Vietnam and Burma, and he’s interested in performing a detailed analysis on the population that lives right on Yunnan’s borders. “We want to do a comparison study and see how fast the virus is disseminated from the border inland to the province,” he says, as the studies could give them insight into how the HIV epidemic may spread in other parts of the country.
It could also have implications for vaccine development. Because the virus is so genetically diverse, researchers must either tailor a vaccine to a particular subtype or create a number of vaccines that can be recombined depending on the variant they’re targeting. Chen, Ho and their associates at ADARC and in China are working hard to produce a vaccine and move it to human trials in Yunnan. Knowing where the different HIV subtypes are dominant can also give them a leg up, letting them test their vaccine in an area where the patients will be more likely to have a viral subtype that matches it.
Citation: Public Library of Science Medicine 3(11): e443 (November 14, 2006)
Credit: Rockefeller University