New methods identify and manipulate 'newborn' cells in animal model of Parkinson's disease
When cells in the brain are lost through disease or injury, neighboring cells begin to divide and multiply, but only a few areas in the brain are able to produce new neurons. Patients with Parkinson's disease suffer degeneration of certain neurons that reside in an area of the brain called the substantia nigra and project into the striatum. Many of the newborn cells in these areas have not been well described because of limitations of methods used to characterize them.
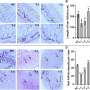
A research team from Cedars-Sinai Medical Center's Maxine Dunitz Neurosurgical Institute and Lund University in Sweden used an engineered virus to deliver a protein that glows green when exposed to blue light (green fluorescent protein) into newborn cells of the striatum in an animal model (rats) of Parkinson's disease. This revealed that no neurons are formed; most of the cells appear to be glial (structural) cells.
To determine if the newborn cells could be manipulated to generate neurons, the researchers delivered into the cells two genes (neurogenin2 and noggin) that are involved in the genesis of neurons. Neither gene had any effect on the ability of newborn striatal cells to form new neurons, but the insertion of noggin greatly increased the number of oligodendrocytes, cells that support neurons.
Dwain Morris-Irvin, Ph.D., a research scientist at Cedars-Sinai's Maxine Dunitz Neurosurgical Institute and Lund University, is available to describe how scientists are using green fluorescent protein and other new approaches in their effort to find cures for Parkinson's disease and other neurodegenerative brain disorders. Morris-Irvin is the first and corresponding author of a recent cover article in Neurobiology of Disease.
"These results may have great potential for studying the effects of viral gene delivery in the attempt to generate new cells for cell replacement therapy in neurodegenerative diseases or for brain repair after injury," Morris-Irvin said. "The success of a 'self-repair' strategy depends on the continued growth of our understanding of complex signaling patterns governing the development of these newborn cells."
Source: Cedars-Sinai Medical Center