Gut organisms could be clue in controlling obesity risk
The international obesity epidemic is widespread, nondiscriminatory, and deadly. But do we really understand all of the factors underlying this alarming trend? The concept of energy balance (energy consumed = energy expended + energy stored) is undeniable, being driven by the first law of thermodynamics. Consequently, there is no contradiction that excessive calorie intake and plummeting levels of physical activity are largely to blame for our ever-expanding waistlines. However, scientists remain baffled as to why some individuals are particularly prone to becoming obese and if there is anything aside from lowering calorie consumption and increasing activity levels that can be done to prevent and/or reverse excessive weight gain in our most at-risk populations.
Physiologists have long known that our intestines are brimming with live bacteria, some of which provide important substances (e.g., vitamin B-12) to their host. However, research conducted over the last decade suggests that these organisms, often referred to as intestinal microbiota, may play a far greater role in human health than previously imagined. One area of intense interest is the possibility that the mix of intestinal bacteria with which we are endowed might directly influence our risk for obesity. Obese individuals tend to have different microbial profiles in their intestines than lean individuals, and scientists have learned that the bacteria common to obesity may metabolize the food we eat in a way that allows us to harvest more calories from it and deposit those calories as fat.
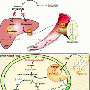
To determine whether altering one's bacterial profile can change obesity risk, researchers from the French Institute for Agricultural Research (INRA) transferred the intestinal bacteria of obesity-prone or obesity-resistant rats into the intestinal tracts of germ-free mice recipients, therefore having no innate gut microbiota. Some animals were fed a regular diet, whereas others were provided unlimited access to a high-fat diet. Food intake and weight gain were monitored for 8 weeks, and intestinal samples were analyzed for a variety of physiologic markers of metabolism and normal feedback mechanisms known to play a role in maintenance of energy balance.
As hypothesized, mice that received intestinal bacteria from obesity-prone animals ate more food, gained more weight, and became more obese than those receiving microbiota from obesity-resistant animals. Animals with microbiota transferred from obesity-prone animals also exhibited changes in intestinal nutrient sensors and gut peptide levels, likely influencing how the animals responded to eating.
The authors' conclusions are three-fold. First, they theorize that obese individuals, when given the opportunity to overeat, may harbor specific gut microbiota profiles that promote excess weight gain. Second, they propose that differences in gut microbes can be related to behavioral changes and increased food intake. Finally, they believe that the mix of microbiota you have may influence your ability to properly sense and respond to a meal. They hope to eventually find ways to manipulate the intestinal microbiota profiles of especially at-risk individuals so that they can more easily maintain a healthy body weight.
As part of the scientific program of the American Society for Nutrition, the nation's leading nutrition research society, results from this study will be presented on April 23, 2012 in San Diego, CA.