Short/long-term analyses deem etanercept safe for psoriasis
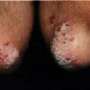
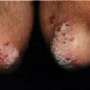
(HealthDay) -- Etanercept treatment is well tolerated for psoriasis, with no indication of dose-related or cumulative toxicity in short- or long-term analyses, according to a study published in the August issue of the Journal of the American Academy of Dermatology.
David M. Pariser, M.D., of Virginia Clinical Research Inc. in Norfolk, and colleagues integrated safety data from short- and long-term etanercept psoriasis trials to determine whether higher doses or extended exposure to etanercept increases the safety risk.
The researchers found that the rate of infectious and noninfectious adverse events (AEs) did not differ significantly between etanercept and placebo treatment groups in short-term analyses (up to 12 weeks of treatment). No dose-related increases in AEs were noted with either short- or long-term (up to 144 weeks) etanercept treatment. Cumulative event rates for serious infections were not significantly different over time or between dose groups. There was no significant difference in the standardized incidence ratios for malignancies excluding nonmelanoma skin cancer. No increase in overall malignancies was noted with etanercept treatment. Congestive heart failure, lymphoma, demyelination, and opportunistic infection were all rare.
"These integrated short- and long-term analyses demonstrated that etanercept was generally well tolerated in a large population of patients with psoriasis without dose-related or cumulative toxicities," the authors write.
Several authors disclosed financial ties to the pharmaceutical industry.
More information:
Abstract
Full Text (subscription or payment may be required)
Copyright © 2012 HealthDay. All rights reserved.