October 18, 2012 report
Researchers use stem cells to show connection between neural cell disruption and Parkinson's disease
(Medical Xpress)—A diverse team of biologists has shown using induced pluripotent stem cells (iPSCs) that a gene mutation that causes malformations in the structure of the nuclear envelope of neural cells, is associated with Parkinson's disease. In their paper published in the journal Nature, they describe how they found iPSC cells taken from Parkinson's patients over time demonstrated the same cell disruption found in neural cells taken from other deceased patient's with the disease. They also found that by introducing a compound known to disrupt the gene mutation, that they could reverse the cell malformation.
Parkinson's disease is a degenerative disorder of the nervous system characterized by shaking, slowness of movement and difficulty walking. Over time most patients succumb to dementia and eventually die. Much research has centered on the disruption and death of dopamine-generating cells as the root cause of the disorder despite evidence that such a disruption would not result in all of the symptoms Parkinson's patient's exhibit. For that reason, researchers have looked to other causes.
In this new effort, the researchers looked at possible reasons for disruption to the nuclear envelope, the thin film that separates the nucleus from the cytoplasm in neural cells. Such disruptions have been associated with Parkinson's but no definitive correlation has been found, until now.
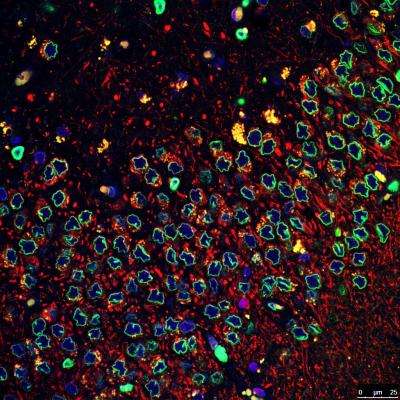
To gain a better understanding of what might be causing such disruptions, the research team obtained samples of induced iPSCs from Parkinson's patients and allowed them to grow in an external environment. They noted that the same disruptions occurred as the iPSCs grew into neural cells, suggesting a genetic cause. Prior research had indicated that a mutation of the LRRK2 gene was connected to Parkinson's disease but no clear indication of the mechanism involved had been found. Testing the cells derived from the iPSCs showed the same mutation, implicating it as a possible cause of the disorder. The researchers also induced the mutation in human embryo stem cells and found that they too developed the same disruption as they grew into neural cells as was found with the iPSCs.
Next the researchers generated a line of iPSCs minus the mutation and found that the cells did not develop the disruptions. They followed that up by adding a chemical compound known to disrupt the mutation to already affected cells and discovered that it prevented them from being disrupted as well.
The researchers don't know why the mutation occurs but believe a new therapy for treating Parkinson's patients might be on the horizon as a result of their research.
More information: Progressive degeneration of human neural stem cells caused by pathogenic LRRK2, Nature (2012) doi:10.1038/nature11557
Abstract
Nuclear-architecture defects have been shown to correlate with the manifestation of a number of human diseases as well as ageing. It is therefore plausible that diseases whose manifestations correlate with ageing might be connected to the appearance of nuclear aberrations over time. We decided to evaluate nuclear organization in the context of ageing-associated disorders by focusing on a leucine-rich repeat kinase 2 (LRRK2) dominant mutation (G2019S; glycine-to-serine substitution at amino acid 2019), which is associated with familial and sporadic Parkinson's disease as well as impairment of adult neurogenesis in mice. Here we report on the generation of induced pluripotent stem cells (iPSCs) derived from Parkinson's disease patients and the implications of LRRK2(G2019S) mutation in human neural-stem-cell (NSC) populations. Mutant NSCs showed increased susceptibility to proteasomal stress as well as passage-dependent deficiencies in nuclear-envelope organization, clonal expansion and neuronal differentiation. Disease phenotypes were rescued by targeted correction of the LRRK2(G2019S) mutation with its wild-type counterpart in Parkinson's disease iPSCs and were recapitulated after targeted knock-in of the LRRK2(G2019S) mutation in human embryonic stem cells. Analysis of human brain tissue showed nuclear-envelope impairment in clinically diagnosed Parkinson's disease patients. Together, our results identify the nucleus as a previously unknown cellular organelle in Parkinson's disease pathology and may help to open new avenues for Parkinson's disease diagnoses as well as for the potential development of therapeutics targeting this fundamental cell structure.
© 2012 Medical Xpress