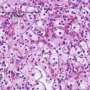
Scientists investigate potential markers for a response to sunitinib in patients with metastatic RCC
Markers such as CA9, CD31, CD34 and VEGFR1/2 in the primary tumours might serve as predictors of a good response to a sunitinib treatment in patients with metastatic clear cell renal cell carcinoma (ccRCC), according to a new study to be presented at the 28th Annual EAU Congress currently on-going in Milan.
"The inactivation of the von Hippel-Lindau gene (VHL) is a common event in ccRCC and finally leads to the induction of HIF1α target genes such as CA9 and VEGF," write the authors. "Besides VEGF, the VEGF and PDGF receptors also play an important role in angiogenesis that is reflected by the microvessel density (MVD).
The tyrosine kinase inhibitor (TKI) Sunitinib targets amongst others the receptors of VEGF and PDGF and is currently one of the standard treatment options for metastatic ccRCC."
According to the researchers, there is currently a lack of predictive biomarkers for a response to TKI treatment. In this study, primary tumours from ccRCC patients, who were later treated with sunitinib, were used for biomarker analyses. In the course of the study, DNA was isolated from cryo-preserved tumour tissue specimens from 20 ccRCC patients for VHL copy number and mutation analyses. Tissue microarrays were prepared from 42 paraffin-embedded malignant and corresponding non-malignant renal tissue specimens.
Immunohistochemical staining of VHL, CA9, PDGFRα, PDGFRβ, VEGFR1, VEGFR2, VEGFR3, CD31 and CD34, VEGFR1, VEGFR2, VEGFR3, CD31 and CD34 was assessed by a scoring system including staining intensity and percentage of stained tumor cells as well as vessel (endothelial) staining. The objective response rate was evaluated according to the RECIST criteria after 3, 6, 9 months and last report (12-54 months) of sunitinib treatment.
Copy number loss and mutation of VHL was observed in 60% and 50%, respectively, with 40% of the cases showing both VHL changes. These VHL gene alterations were accompanied by a reduced VHL protein staining and an increased CA9 score.
After 9 months, 45% of the patients responded to sunitinib treatment. Their response was associated with low Fuhrman grade of the primary tumour (p<0.05). The vessel staining of VEGFR1/2 was elevated in patients with a response after last report.
Patients with a response after 6 months exhibited an increased MVD and CA9 staining in the primary tumour (p<0.05) and their estimated median progression free survival (12 mo vs. 8 mo; p<0,001) and median overall survival (45 mo vs. 21 mo; p<0.001) was longer than for non-responders. High CA9 and low PDGFRα; protein expression were associated with longer overall survival (p<0.01).
More information: J. Dornbusch, et al., "Potential predictive markers for a response to sunitinib and their association with survival of patients with metastatic renal cell carcinoma," Abstract Nr: 984; 28th Annual EAU Congress, 15 to 19 March 2013; Milan, Italy.