Uncleaned cells mean weak muscles
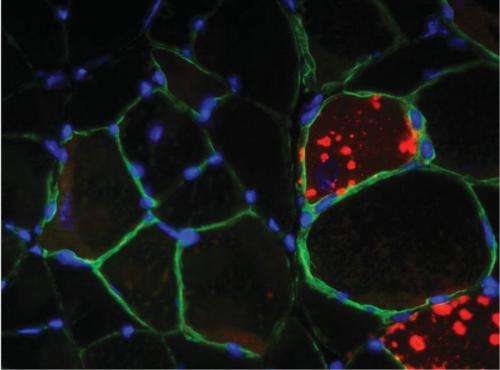
The protein complex mTORC1 promotes muscle growth. However, should this complex remain constantly active, it impairs the ability of the cells to self-clean, causing myopathy. Scientists working with Markus Rüegg, Professor at the Biozentrum, University of Basel, describe the exact mechanism involved in the current issue of the scientific journal Cell Metabolism.
Similarly to parts in a machine, individual components of a cell wear out with time. For a cell to remain healthy, malfunctioning components and waste products must be regularly disposed of or recycled. A cellular self-cleaning process, called autophagy, is responsible for this. However, the capacity for self-renewal decreases with age and participates in a wide range of age-related diseases such as cancer, heart disease and muscle weakness. In this process, the growth regulator, mTORC1, plays a primary role. The exact relationship has now been discovered by Markus Rüegg's team from the Biozentrum of the University of Basel, together with scientists from the Department of Biomedicine.
Muscle weakness due to overactive growth regulator
Until recently, it was assumed that the protein complex mTORC1 in the skeletal muscle plays a key role in growth regulation but not in the process of autophagy. Rüegg and his team of scientists have been able to refute this widely accepted assumption. In the current study, they investigated the cellular processes in skeletal muscle of mice, in which mTORC1 was permanently activated. Particularly in aging mice, the scientists observed a progressive myopathy, which could be ascribed to impaired autophagy. Interestingly, the researchers could reverse the symptoms by administering rapamycin. The muscle function of the mice returned to normal. Rapamycin is a substance that inhibits mTORC1, thereby promoting cell self-cleaning.
Counteracting muscle breakdown
According to these findings, mTORC1 plays a major role in tightly coordinating the mechanism of autophagy, maintaining the balance between muscle growth and breakdown. The scientists suspect that an overactive mTORC1 complex may also contribute to the development of the age-related muscle weakness seen in man. Therefore, a closer examination of the mTORC1 regulation system in the context of aging may provide new therapeutic approaches for the counteracting of the muscle weakness.
More information: Perrine, C. et al. (2013) Sustained activation of mTORC1 in skeletal muscle inhibits constitutive and starvation-induced autophagy and causes a severe, late-onset myopathy, Cell Metabolism; Published online April 18, 2013. dx.doi.org/10.1016/j.cmet.2013.03.015














