New inhibitor blocks the oncogenic protein KRAS
One of the major goals in the development of anti-cancer treatments is to find an inhibitor effective against the oncogenic protein known as KRAS. Despite decades of active agent research, efforts to intercede in this protein's cancer-promoting effect have not been successful. KRAS only becomes fully functional once it is anchored in the cell membrane. Therefore, the location of KRAS in the cell is closely connected with its cancer-promoting effect. In a new study, researchers at the Max Planck Institute of Molecular Physiology in Dortmund have now discovered how the transport of KRAS to the cell membrane can be interfered with by a new type of inhibitor. The approach that Deltarasin takes as an inhibitor is not directed against KRAS itself as previous approaches have been, but instead against the KRAS transport protein PDEδ. These research results open up approaches to cancer therapy, especially for the most deadly of all cancers, cancer of the pancreas, which is often detected at such a late stage that therapies can no longer counter it.
The protein KRAS is a molecular switch that under normal conditions regulates cell growth and cell division. Mutations in this protein lead to an unregulated, chronic "on" condition and finally to the formation of tumours through continuous cell growth. As every third tumour exhibits a mutation in a RAS protein, these have been the focus of inhibitor research for decades. The KRAS protein occupies a central position of interest. Mutated KRAS proteins are found in a large proportion of all tumour cells from pancreatic cancer patients, for example. Up to now however, it has not been possible to develop a medication against hyperactive KRAS mutations, so that the probability of recovering from pancreatic cancer continues to be minimal.
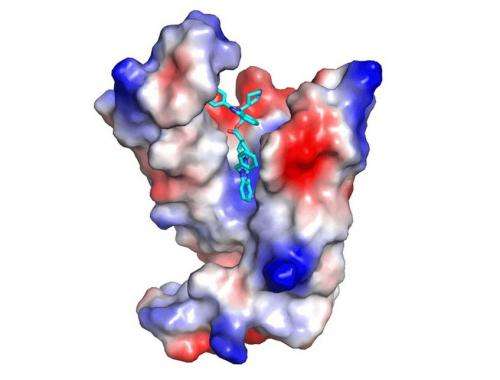
When developing treatments against very dangerous diseases like pancreatic cancer, a small organic molecule is often needed that binds to the surface of a protein like KRAS. The precise fit of the molecule to the protein surface is especially important in this approach. This recognition process is comparable to the functioning of a key for a lock. For decades, the pharmaceutical industry has committed large sums to the search for a compatible molecular key to KRAS, unfortunately without success.
Instead of continuing to search for a key to KRAS itself, the MPI researchers went another route. In their new work they used knowledge from their previous studies in which they identified PDE? as the transport protein for KRAS. By means of the most modern high-throughput technology, the researchers were able to find the first weak inhibitor substances to this auxiliary protein PDE?. In a cooperative effort between chemists and structural biologists, these were subsequently adapted to improve the fit to PDE?.
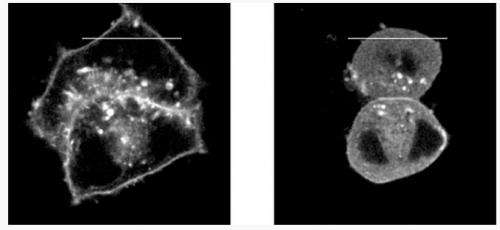
The most effective inhibitor, named deltarasin, was tested by cell biologists for its effectiveness in relation to the localisation of KRAS within cultured human cells. They were able to show that KRAS no longer remained at the plasma membrane, but instead was distributed throughout the cell. Subsequently, they investigated the growth of tumour cells whose survival depends on KRAS mutations compared to control cells. After treating the cells with the inhibitor, they could observe a die off of what are known as the KRAS-dependent tumour cells, while control cells without KRAS mutations continued to grow.
At the end of the study, deltarasin was tested as an inhibitor in mice that had previously received injections of human tumour cells with KRAS mutations. Here as well, the deltarasin was effective as an inhibitor in sharply reducing tumour growth in comparison to a placebo.
Deltarasin as an optimised inhibitor is thus a milestone in the area of active substance research and has already inspired many pharmaceutical companies to work on tumours having KRAS mutations.
More information:
Zimmerman, G. et al. (2013). Small molecule inhibition of the KRAS-PDE? interaction impairs oncogenic KRAS signalling.
Nature, 2013 May 30;497(7451):638-42. DOI: 10.1038/nature12205