Re-thinking cancer treatment
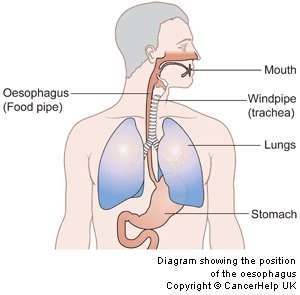
A new treatment approach for tackling cancer of the food pipe (oesophagus) has been developed by researchers at Cardiff and Velindre NHS Trust.
Oesophageal cancer is widely considered to be one of the most difficult cancers to treat, with 8000 new diagnoses every year, equating to over 150 people a week.
Supported by Cancer Research UK (CRUK) funding, researchers are now trialling a combination of ground-breaking radiotherapy techniques and two different chemotherapy drugs before surgery in an effort to ultimately improve survival rates in patients.
"This is a really exciting study that aims to use the most effective drugs combined with cutting edge technologies to find new avenues to treatment," said Dr Tom Crosby, Clinical Director of Velindre Cancer Centre, who is leading the study.
"Patients' chances of beating this type of cancer are often hampered by late diagnosis, which means doctors are left treating the condition in its advanced stages – a new approach is needed if we are to stand a fighting chance of stopping its spread and killing it completely."
For patients who are suitable for surgery, doctors can either treat patients with chemotherapy or with a combination of chemotherapy and radiotherapy (chemo-radiotherapy) before their operation. Recent studies have shown that chemo-radiotherapy, delivered with new radiation technology and different combinations of chemotherapy drugs, may be safer and more effective than chemotherapy alone.
Following the trial, patients will have their tumour surgically removed and will be monitored by a research team for up to 12 months. Doctors will look at the side-effects experienced by the patients, how long patients remain free from cancer and whether patients show spread of the cancer to other parts of the body.
The trial, run by the Wales Cancer Trials Unit (WCTU), located within the School of Medicine involves 25 hospitals and 85 patients from Wales and England and is sponsored by Velindre NHS Trust.
"This clinical trial has just opened and has recruited its first few patients. It will give us the evidence to determine whether to investigate this new treatment in a larger number of patients which could show a benefit to patients in terms of survival and could ultimately change routine practice in the UK" said Dr Gareth Griffiths, Director of Cancer Research UKs Wales Cancer Trials Unit at Cardiff.