Nontoxic hydrogel for breast cancer treatment
A nontoxic hydrogel developed by the A*STAR Institute of Bioengineering and Nanotechnology and IBM Research offers a new way forward for breast cancer therapeutics.
Breast cancer is the most common form of invasive cancer affecting women worldwide. Treatment usually involves surgery followed by a course of either chemotherapy, radiotherapy or hormone therapy designed to reduce the risk of the cancer's recurrence. In recent years, however, targeted drugs have increasingly become an effective option to fight the disease.
One such drug is Herceptin, also known as trastuzumab, a monoclonal antibody that can slow or even halt tumor growth in patients with human epidermal growth factor receptor 2 (HER2)-positive breast cancer—a particularly fast-growing form of the disease that affects one in four patients. Typically, the drug is administered at a clinic through an intravenous drip, a process that can take up to 90 minutes. A major drawback of this method of delivery is that without frequent follow-up doses, Herceptin loses its effectiveness. Therefore, patients are commonly required to visit the clinic on a weekly basis.
Now, a team of researchers from the A*STAR Institute of Bioengineering and Nanotechnology (IBN) and IBM Research led by Yi Yan Yang and James Hedrick has developed a more efficient way of delivering Herceptin to breast cancer cells. The approach promises to improve the effectiveness of treatment for the HER2-positive form of the disease. Drawing on IBN's expertise in the development of novel biomaterials, the researchers recognized the advantages of using a hydrogel—comprising 96 per cent water and a uniquely designed polymer—as a carrier for Herceptin in the body.
The hydrogel is nontoxic, biodegradable and can be injected under the skin without causing an inflammatory response. By varying the composition and concentration of the polymer, the researchers were able to fine-tune the hydrogel's properties to ensure a slow and sustained release of the anticancer drug, thus increasing the efficiency of delivering the drug to the target site. Correspondingly, use of the hydrogel for drug delivery has the potential to reduce the frequency at which patients need to be injected with Herceptin from once a week to once every four weeks.
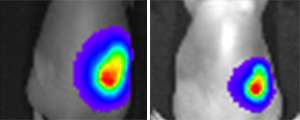
Recently, the researchers conducted studies in mice, which confirmed that their Herceptin-loaded hydrogel delivers the drug efficiently, before it degrades within 6 weeks. They found that 4 weeks after injection, tumors in the mice had decreased in size by as much as 77 per cent. The researchers say that their next goal will be to conduct clinical trials in humans, in conjunction with industrial partners.
Other teams at IBN are also exploring the innovative use of hydrogels for controlled-release drug delivery to tackle different types of cancer (see Greater anticancer potency with less risk). The institute has always cultivated a multi-pronged approach toward cancer research, notes Jackie Y. Ying, professor and executive director at IBN.
"Our multidisciplinary research teams are working with various industrial, clinical and academic partners to develop new materials and tools to improve cancer diagnosis and treatment," she adds. "This latest breakthrough with our long-term partner IBM Research promises more efficient administration of anti-cancer drugs and more effective treatment of breast cancer, which we hope will benefit breast cancer patients worldwide."