Blocking immune system protein in mice prevents fetal brain injury, but not preterm birth
An inflammatory protein that triggers a pregnant mouse's immune response to an infection or other disease appears to cause brain injury in her fetus, but not the premature birth that was long believed to be linked with such neurologic damage in both rodents and humans, new Johns Hopkins-led research suggests.
The researchers, reporting online March 5 in the American Journal of Reproductive Immunology, also say they found that an anti-inflammatory drug that is FDA-approved for rheumatoid arthritis and is believed to be safe for humans to take during pregnancy halted the brain injury in mouse offspring. The treated mice exposed prenatally to the inflammatory protein were still born before reaching full term, but without neurologic damage.
"Down the road, we hope to be able to treat pregnant women whose babies are at risk for this type of neurologic damage and prevent it from happening," says study leader Irina Burd, M.D., Ph.D., an assistant professor of gynecology and obstetrics and neurology at the Johns Hopkins University School of Medicine and director of the Integrated Research Center for Fetal Medicine.
Roughly 500,000 infants are born preterm in the United States each year, accounting for close to 12 percent of all live births. Such babies have increased rates of illness and death, but they are also at higher risk for cognitive and neurobehavioral disorders as they grow older. Studies have identified an association between maternal infection and inflammation and preterm birth, in addition to the development of cerebral palsy and neuropsychiatric conditions such as schizophrenia and autism.
In research designed to identify the exact mechanisms behind preterm birth and fetal brain injury, Burd says she and her term were surprised to learn that they may have different pathways and origins.
"We didn't expect to stop brain injury in our mice without also stopping preterm birth, the impact of which extends beyond the brain," she says.
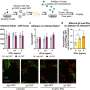
In the study, one group of pregnant mice got saline injections into the womb, while another group got injections of lipopolysaccharide (LPS), a toxin meant to generate the kind of inflammatory effects of E. coli bacteria without the presence of the germ itself. A third group first got an injection of a compound known to block the effects of interleukin-1 beta (IL-1), a protein involved in activating the inflammatory immune response and shown in previous research to be a key player in modulating fetal brain injury following exposure to inflammation in the womb. Thirty minutes after getting the IL-1 blocking agent, those same mice got an LPS injection.
As expected, the saline-only group delivered no preterm offspring. In the other groups, 80 to 95 percent of the litters were delivered prematurely. Meanwhile, mice that got the IL-1 blocking drug—sold in the United States under the trade name Kineret—did not suffer the harmful neurological effects of the inflammatory response experienced by the group that received only the LPS. The researchers say that blocking IL-1 halted a cascade of inflammatory responses that would otherwise lead to the activation of neuronal nitric oxide synthase in the fetal brain, an enzyme implicated in neurotoxicity.
"Those mice that got the drug while pregnant still delivered early, but the drug prevented neuronal injury in their offspring," Burd says.
The brains of the mice that got only LPS had fewer dendrites attached to their neurons. Dendrites are fingerlike extensions of the neuron that form with others to create the synapses, or the circuits that help neurons communicate with one another. Those that received the IL-1 blocker had a similar number of dendrites to those mice that only got saline.
Burd cautions that they are still a long way from the use of the drug in humans.