Dual-action chemical agents improve a high-resolution and noninvasive way to detect cancer
Concerns over toxicity have constrained the clinical application of photoacoustic imaging—a new experimental technique used to detect tumors lurking among healthy cells. A*STAR researchers from the Singapore Bioimaging Consortium and an international team have now discovered how to improve both the safety and the tumor-locating efficacy of photoacoustic imaging using 'photosensitizer' contrast agents.
Unlike typical optical measurements, photoacoustic imaging can probe deep into tissue by using ultrasonic waves generated by laser light pulses. This hybrid method produces strikingly clear pictures of complex biosystems, such as vascular tumor networks in organs and tissues. However, clinical applications of photoacoustic imaging have been limited by safety concerns surrounding 'contrast agents'—light-absorbing molecules injected into cells to enhance imaging resolution.
Photosensitizers are molecules that play key roles in a cancer treatment known as photodynamic therapy. These nontoxic substances, which are normally based on conjugated aromatic ring systems, specifically bind to malignant cells while leaving healthy tissue untouched. Exposing a photosensitizer to laser light causes it to produce excited-state oxygen molecules, which react with diseased cells to kill them. The photosensitizer is then rapidly eliminated from the target tissue.
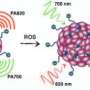
Photosensitizers have often been overlooked as diagnostic imaging aids because, after laser excitation, their fluorescence output is not penetrating enough for deep-tissue imaging. But Malini Olivo and his team realized that the poor fluorescence efficiency of excited-state photosensitizers could prompt them to relax though thermal conversions instead—conditions that might favor production of acoustic pressure waves for photoacoustic imaging. To achieve this, the researchers needed to find photosensitizers that can absorb light at the near-infrared wavelengths used for deep-tissue imaging.
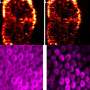
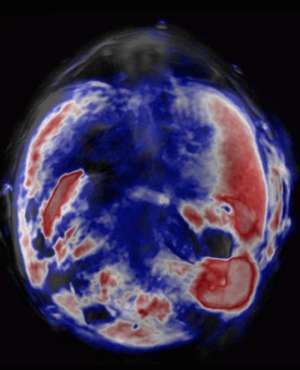
After testing different photosensitizers, the team selected a promising substance known as zinc phthalocyanine (ZnPc) to inject into living mice with tumors. Experiments confirmed their hypothesis: the photosensitizers produced strong photoacoustic signals as they passed through vital organs and zeroed in on the tumor cells. Within ten minutes of injection, signals from the ZnPc contrast agents illuminated the precise position of diseased tissue (see image). The ZnPc cleared harmlessly from the mice within one day, resulting in a low overall toxicity.
"Many photosensitizers have already been widely studied in clinical trials, and so using them in photoacoustic imaging offers great potential as an alternative diagnostic technique," says Olivo. "Combining the preferential tumor uptake and high photodynamic therapy efficacy of photosensitizers with photoacoustic imaging can help achieve long-term monitoring of cancer progression and therapy under in vivo conditions."
More information: Ho, C. J. H., Balasundaram, G., Driessen, W., McLaren, R., Wong, C. L. et al. "Multifunctional photosensitizer-based contrast agents for photoacoustic imaging." Scientific Reports 4, 5342 (2014). dx.doi.org/10.1038/srep05342