Tumor segmentation software receives 510k clearance from FDA
Columbia University's imaging software that facilitates 3-D lung tumor segmentation, licensed to Varian Medical Systems, has been incorporated into the Smart Segmentation module of Varian's Eclipse treatment planning system and has received FDA 510k clearance.
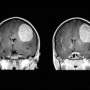
Three-D segmentation of CT and MR images provides a reliable way to identify various organs and tumors within anatomical images, such as the brain, lymph node, lung, liver, spleen, and kidneys. Determining organ and tumor contours and volumes (including those of primary and metastatic tumors) before, during, and after treatment can be challenging. Accurate and efficient segmentation and characterization of these diverse structures is necessary to enable noninvasive assessments in clinical practice as well as in radiation treatment planning.
"Our organ- and tumor-specific segmentation programs have been developed in response to the growing demand for quantitative imaging techniques and extensively field tested for clinical purposes, with very promising and encouraging results. They can be readily integrated into routine clinical workflow. We are pleased that Varian Medical Systems has used these programs as part of its product and has received regulatory clearance from the FDA, as this will pave the way for broader, global access to tumor segmentation software," said Lawrence Schwartz, MD, the James Picker Professor of Radiology and chair of the Department of Radiology, Columbia University Medical Center.
"Working closely with physicians, we developed our advanced computer algorithms with the aim of improving accuracy and efficiency in cancer diagnosis, treatment planning, and response assessment. We are pleased to note an increasing demand for licenses to our proprietary software," said Binsheng Zhao, DSc, professor of radiology and director of the Computational Image Analysis Laboratory. "In the Lab, we continue to innovate and have developed new advanced methodologies to address these various imaging needs."