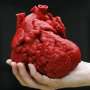
Surgeons use 3-D printed model of heart to treat patients with disorders
An experimental 3-dimensional printed model of the heart may help surgeons treat patients born with complicated heart disorders, according to research presented at the American Heart Association's Scientific Sessions 2014.
Most heart surgeons use 2D images taken by X-ray, ultrasound and MRI for surgical planning. However, these images may not reveal complex structural complications in the heart's chambers that occur when heart disease is present at birth (congenital heart defects), as opposed to developing later in life within a structurally normal heart.
But with standard 2D images as a guide, doctors now can build a detailed 3D model of the heart from various materials, such as plaster or ceramic, to reveal even the most complicated structural abnormalities.
"With 3D printing, surgeons can make better decisions before they go into the operating room," said Matthew Bramlet, M.D., study lead author and assistant professor of pediatric cardiology and director of the Congenital Heart Disease MRI Program at the University of Illinois College of Medicine in Peoria. "The more prepared they are, the better decisions they make, and the fewer surprises that they encounter.
"When you're holding the heart model in your hands, it provides a new dimension of understanding that cannot be attained by 2D or even 3D images. What once was used to build trucks, we're using now to build models of hearts."
Researchers used an inexpensive plaster composite material to create heart models of a 9-month-old girl, 3-year-old boy and a woman in her 20s all of whom had complex congenital heart defects. After studying the models and traditional images, surgeons successfully repaired severe heart abnormalities in all three patients.
"You could see that if you make this compromise here, you could fix this problem, and go from a
single-ventricle to a two-ventricle repair," Bramlet said. "That is the difference, potentially, between a life expectancy of two to three decades, to four, five or six decades."
Researchers caution that this was a small study and 3D printing is still an emerging technology that is not approved by the Food and Drug Administration. The University's collaborator, the Jump Trading Simulation and Education Center in Peoria, made the printer available for the study.