A protein on which two molecular pathways converge could be the key to new cancer therapies
Two molecular pathways—one that causes cancer and one that protects against it—compete to control cellular levels of one protein, according to a new study by A*STAR researchers and colleagues.The finding highlights a central role for the protein in cancer, and could lead to new therapies.
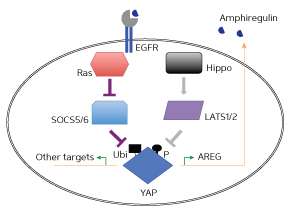
The Hippo pathway regulates the activity and levels of a protein called Yes-associated protein (YAP), and is known to protect against cancer, making it a potential target for cancer drugs. Now, work led by Xin Hong and Hung Nguyen in Stephen Cohen's research group at the A*STAR Institute of Molecular and Cell Biology has shown that the Ras pathway, which is known to have a role in the development of tumors, competes with the Hippo pathway to regulate the activity and levels of YAP.
Previous work by the same international research team showed that reducing cellular levels of proteins called suppressors of cytokine signaling 5 (SOCS5) enhanced the ability of the Ras protein to transform normal cells into cancer cells. Building on these findings, the researchers directly manipulated levels of SOCS proteins in cultured human epithelial cells, and showed that depletion of these proteins had the same effect as activation of Ras: that is, increased activity and levels of YAP that led to an increased ability of normal cells to transform into cancer cells.
The team went on to show that SOCS6 normally forms a complex with YAP that is targeted for degradation. Depleting the SOCS proteins therefore prevents formation of this complex and reduces the degradation of YAP, which leads to increased cellular levels of the protein. This process is in contrast to the Hippo pathway, which encourages tagging of YAP for degradation, thereby preventing its levels in cells from increasing.
"Ras pathway activity promotes YAP stability, acting in opposition to the Hippo pathway, which promotes YAP turnover," summarizes senior investigator Cohen. "It seems that all roads lead to YAP."
"Our study also suggested that amplification or activation of YAP could act as a backup mechanism to support tumor growth if Ras activity is shut off," adds Hong. "This suggests that YAP activity might contribute to drug resistance when Ras inhibitors are applied to cancer cells."
The position of YAP at the interface of two pathways that are important in cancer makes it a prime target for new therapies.
"The next stage in our research is to identify druggable targets that regulate YAP activity in desirable ways," concludes Cohen.
More information: Hong, X., Nguyen, H. T., Chen, Q., Zhang, R., Hagman, Z. et al. "Opposing activities of the Ras and Hippo pathways converge on regulation of YAP protein turnover." EMBO Journal 33, 2447–2457 (2014). dx.doi.org/10.15252/embj.201489385