New tool helps identify lung cancer patients who will respond to immune therapies
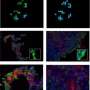
A Yale-led team of researchers has developed a new assay, or investigative tool, to measure the anti-tumor immune activity in non-small cell lung cancer tumors that could lead to a more accurate determination of which patients will respond to immune therapy drugs. Findings from the study were published in the Journal of the National Cancer Institute.
The assay simultaneously measures subpopulations of tumor-infiltrating lymphocytes (TILs), a type of white blood cell that attacks tumors. The presence of high amounts of TILs in tumors is associated with better treatment outcomes. The new method differs from existing immune-measuring assays in that it is objective, quantitative, and reproducible, said the paper's first author, Dr. Kurt Schalper, associate research scientist in Yale School of Medicine and director of the Translational Immuno-oncology Laboratory at the Yale Cancer Center.
"The strength of our method is that it allows simultaneous measurement of different TIL subtypes and removes the subjectivity from the process by using automated scoring. In addition, this assay can assess TILs activity in different tumor compartments," Schalper said. "We believe that this method could help determine which patients are more likely to benefit from new immune checkpoint therapies."
Accurately measuring the properties of the anti-tumor immune responses could lead to novel immunostimulatory therapies, such as anti CTLA-4 (ipilimumab) and PD-1/PD-L1 (nivolumab), which have shown unprecedented clinical responses in diverse cancers. Future efforts will be focused on validation of these measurements and their integration into routine cancer patient assessment.
More information: "Objective Measurement and Clinical Significance of TILs in Non–Small Cell Lung Cancer." JNCI J Natl Cancer Inst (2015) 107 (3): dju435 DOI: 10.1093/jnci/dju435