Sleep apnea therapy treats patients through upper airway stimulation
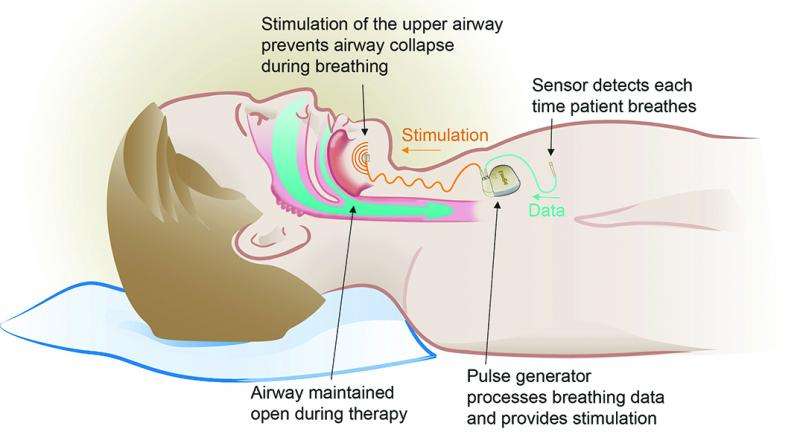
A state-of-the-art implant designed to treat obstructive sleep apnea (OSA) is now being offered at NewYork-Presbyterian/Weill Cornell Medical Center. The device is the first of its kind approved by the U.S. Food and Drug Administration and is an alternative to continuous positive airway pressure (CPAP), the current standard of care for OSA.
Some 18 million Americans have OSA, according to the National Sleep Foundation. The condition occurs when the tongue and other soft tissues relax during sleep and obstruct the airway. While CPAP masks are effective when used properly and consistently, they face high rates of non-compliance. Studies have shown that many patients find CPAP masks uncomfortable and inconvenient to wear at night before going to sleep.
"The implantable upper airway stimulation device has revolutionized our treatment approach for CPAP-intolerant patients with OSA," said Dr. Maria Suurna, a sleep surgery specialist at NewYork-Presbyterian/Weill Cornell and assistant professor of otolaryngology/head and neck surgery at Weill Cornell Medical College. "This is the most exciting alternative therapy for patients who have struggled for years to effectively manage their OSA. Now, we can implant this small device with little discomfort to the patient. This innovative procedure has been very successful in controlling airway obstruction and snoring during sleep."
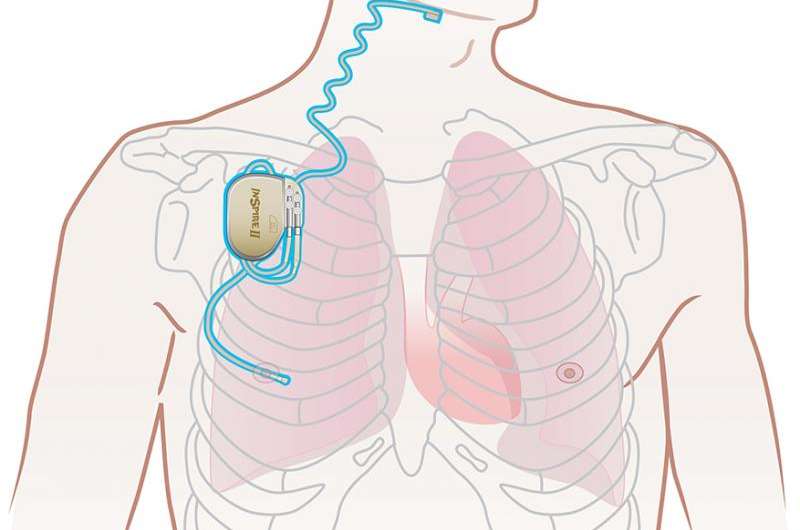
The implant, manufactured by Inspire Medical Systems, works with a person's natural breathing process. The device is inserted under the skin in the upper chest, and delivers mild stimulation to key airway muscles, keeping the airway open during sleep. The patient activates the device with an external remote control at night and can turn it off upon waking in the morning.
OSA poses a wide range of health risks to patients. The condition's common side effects include daytime sleepiness, depression, weight gain and diminished quality of life. In clinical trials, the implant reduced apnea and hypopnea (shallow breathing) by 68 percent, and 85 percent of bed partners reported little to no snoring in their partner 12 months after activation. In addition, patients reported significant improvements in daytime functioning.
"While CPAP treatment can often help people with OSA, there is still a great need for alternative treatment for patients who have not been able to tolerate CPAP," said Dr. Ana Krieger, medical director of the Weill Cornell Center for Sleep Medicine at NewYork-Presbyterian Hospital and an associate professor of clinical medicine, neurology and genetic medicine at Weill Cornell Medical College. "This new implant will allows us to offer a new treatment approach for patients who have been suffering for many years. This patient-centered approach is a major breakthrough in addressing OSA, which is related to a number of other, more serious conditions."
NewYork-Presbyterian/Weill Cornell is currently offering the procedure and device to patients who have OSA and have not been able to use a CPAP mask.