Researchers find key player in diabetic kidney disease through power of metabolomics
Tapping the potential of metabolomics, an emerging field focused on the chemical processes of metabolism, researchers at University of California, San Diego School of Medicine have identified a new and pivotal player in diabetic kidney disease.
The study, published online July 22 in the Journal of the American Society of Nephrology, also clarifies a central mechanism of action in diabetic kidney disease that is generating considerable excitement among researchers and the biopharmaceutical community. The mechanism, involving the NADPH (nicotinamide adenine dinucleotide phosphate) oxidase (NOX) proteins, NOX1 and NOX4, is now the subject of a phase II clinical trial for the treatment of diabetic kidney disease.
"Our study further illuminates the role of NOX proteins, in particular NOX4, in mediating diabetes-associated kidney dysfunction and identifies fumarate, a product of the TCA (tricarboxylic acid cycle) and urea cycles, as a key link in the metabolic pathways underlying diabetic kidney disease," said senior author Kumar Sharma, MD, a professor of medicine and director of the Center for Renal Translational Medicine at UC San Diego School of Medicine. By pinpointing fumarate, Sharma added, the research team has also discovered a new, and potentially better, biomarker for diagnosing and monitoring chronic diabetic kidney disease. Young-Hyun You, PhD, a project scientist in the Center for Renal Translational Medicine, was first author on the study.
Diabetic kidney disease is the leading cause of end-stage kidney disease, the eighth leading cause of death in the United States and a major risk factor for cardiovascular disease. An estimated 26 million American adults have chronic kidney disease, often requiring dialysis or a kidney transplant.
Previous studies by UC San Diego researchers, including Sharma and David Brenner, MD, vice chancellor for Health Sciences and dean of the School of Medicine, and other research groups around the world, have shown that NOX 4 plays an important role in several progressive fibrotic and inflammatory diseases, including liver fibrosis and kidney disease. Sharma said their new study sought to delve deeper into how the NOX proteins cause problems in the kidneys and used the analysis of urine metabolites to better understand the process.
Measuring numerous metabolites to detect and assess diabetic kidney disease is a recent diagnostic advance, said Sharma. Metabolites are the small molecules involved in or produced through metabolic processes in cells. As the by-products of metabolic processes, metabolites serve as lingering chemical fingerprints that researchers can analyze to learn more about how the body functions in health and disease. More than 10,000 distinct metabolites are estimated to reside in the human body and encompass a vast spectrum of chemical types.
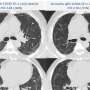
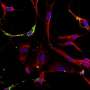
In the study, the researchers tested mice engineered to overproduce NOX4. "We identified that overproduction of NOX4 in kidney cells, called podocytes, produced many of the features of diabetic kidney disease," said Sharma. The diabetic mice were then given a compound known as GKT137831, a NOX 1/4 inhibitor developed by Swiss pharmaceutical Genkyotex SA, and presently in phase II clinical trials for diabetic kidney disease, also known as diabetic nephropathy. The results of that trial are expected to be announced this year.
The researchers analyzed urine metabolites in the mice and found interesting activity surrounding one particular metabolite. "Many of the TCA metabolites were elevated in the urine of untreated diabetic mice, but only fumarate showed a dose-dependent reduction with the administration of the NOX1/4 inhibitor," Sharma said.
Exploring further, the group found that a key enzyme called fumarate hydratase was reduced in both the human and mouse diabetic kidney, an action they believe may contribute to the elevation of fumarate. High levels of fumarate were found to be "a very powerful regulator of two important pathways that have been linked to diabetic kidney disease, hypoxia inducible factors (HIF-1?) and transforming growth factor-beta (TGF-?)," said Sharma.
Fumarate also stimulated endoplasmic reticulum stress, apoptosis of cells and matrix gene expression - all cellular processes previously shown to contribute to fibrosis, the scarring activity underlying diabetic kidney disease. "We found that administration of the NOX1/4 inhibitor GKT137831 had the downstream effect of reducing fumarate accumulation and fibrotic activity in the kidney," said Sharma.
As a measurable urine metabolite, Sharma said fumarate could serve as a new biomarker for diabetic kidney disease and could prove more effective than the urine albumin protein, which currently serves as a major indicator of kidney disease. "Using fumarate as a biomarker could not only indicate the presence of kidney disease, but also indicate the activity of a NOX4-inhibitor drug, since fumarate levels are tied to NOX4 inhibition," he said. "You could gain the ability to decide how much of the drug to give by measuring its effectiveness (through fumarate levels) in individual patients," he said.
Sharma said next steps will be to further explore urine fumarate levels in human diabetic kidney disease.