October 13, 2015 report
Optogenetic pacing possible alternative to electrical pacemakers
(Medical Xpress)—A team of researchers with Lehigh University in the U.S. has found via experimentation with fruit flies that optogenetic pacing might one day be a viable alternative to electrical pacemakers in humans. In their paper uploaded to the open access site Science Advances, the team describes how the procedure works and why they believe it is superior to traditional pacemakers.
Pacemakers are cigarette pack-sized devices that are implanted under the skin of heart patients to assist or take over the process by which the heart constricts to cause blood to pump. Several ailments can cause erratic heart rates which can lead to poor health or death if not treated properly. Traditional pacemakers send small electrical shocks straight to the heart, causing it to constrict in a regular fashion. But this approach also has its drawbacks, e.g. the need for surgery to implant a device, damage to non-heart tissue, etc. For that reason, scientists continue to look for a better way to get the job done. In this new effort, the team in Pennsylvania looked to using optogenetic pacing, which is where genes in heart muscle cells are modified to cause tissue to react to light and then a laser is used to stimulate the heart into beating at a regular pace.
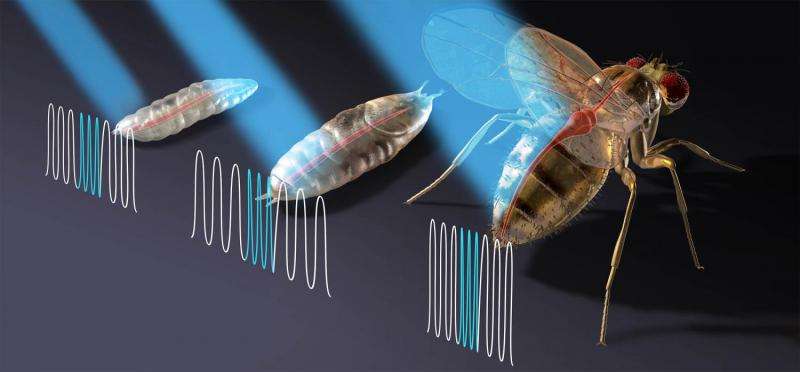
In their experiments, the researchers used Drosophila melanogaster (the common fruit fly) as a stand in for humans, because prior research has shown that approximately 75 percent of heart disease related genes are shared between the two species, and because they have been used in a wide array of heart related research efforts, which means their anatomy and behavior are reasonably well understood.
The team began by developing transgenic flies that expressed the cation channeled protein channelrhodopsin-2 in just their hearts and then used a laser to cause light pulses to pass through the thin walls of the body and to the heart, causing it to beat at a specified regular interval. The team tested the technique on larva, pupa and adults and report that the device worked as envisioned for all stages of a fly's life and did not cause any damage to heart or other tissue. To make sure the device was working as intended the team monitored the heart using a high speed camera.
The technique cannot yet be tested on humans of course because of the need for gene modification—also there is the problem of light reaching through a much thicker chest cavity. Still the team is optimistic that one day a way will be found to use the technique (perhaps using infrared light) providing heart patients with a better option.
More information: Optogenetic pacing in Drosophila melanogaster, Science Advances 09 Oct 2015: Vol. 1, no. 9, e1500639 , DOI: 10.1126/sciadv.1500639
Abstract
Electrical stimulation is currently the gold standard for cardiac pacing. However, it is invasive and nonspecific for cardiac tissues. We recently developed a noninvasive cardiac pacing technique using optogenetic tools, which are widely used in neuroscience. Optogenetic pacing of the heart provides high spatial and temporal precisions, is specific for cardiac tissues, avoids artifacts associated with electrical stimulation, and therefore promises to be a powerful tool in basic cardiac research. We demonstrated optogenetic control of heart rhythm in a well-established model organism, Drosophila melanogaster. We developed transgenic flies expressing a light-gated cation channel, channelrhodopsin-2 (ChR2), specifically in their hearts and demonstrated successful optogenetic pacing of ChR2-expressing Drosophila at different developmental stages, including the larva, pupa, and adult stages. A high-speed and ultrahigh-resolution optical coherence microscopy imaging system that is capable of providing images at a rate of 130 frames/s with axial and transverse resolutions of 1.5 and 3.9 μm, respectively, was used to noninvasively monitor Drosophila cardiac function and its response to pacing stimulation. The development of a noninvasive integrated optical pacing and imaging system provides a novel platform for performing research studies in developmental cardiology.
© 2015 Medical Xpress