Vascular surgery research brings new options to high-risk stroke patients
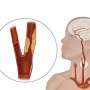
The carotid arteries supply oxygenated blood to the brain. Every year, more than 300,000 people in the United States are diagnosed with blockages, or plaques, in their carotid artery. When these arteries become blocked due to plaque build-up, an individual is at high risk for a stroke if the plaque ruptures and flows to the brain. Many individuals can manage carotid artery disease with medications and lifestyle changes. However, many may require carotid artery revascularization surgery to repair the blockage in the artery. Carotid endarterectomy has been the gold standard of treatment with carotid stenting reserved for those who are high risk for open surgery. While largely effective in some patients, carotid stenting from the transfemoral route has been found to carry a higher risk of stroke, with a significant amount of this risk felt to be related to the approach.
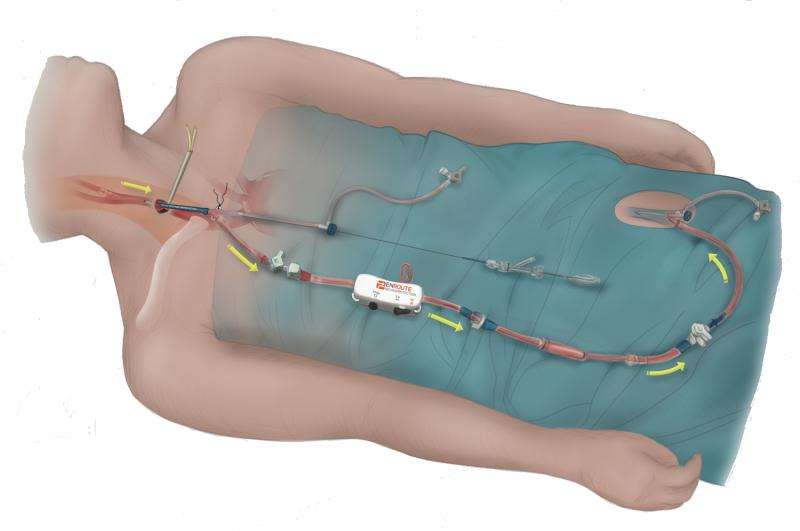
The FDA recently approved a new Transcarotid Artery Revascularization (TCAR) procedure that utilizes the ENROUTE Transcarotid Neuroprotection and Stent System from Silk Road Medical. The TCAR procedure offers patients a potentially safer method of carotid stenting through a small cut down at the base of the neck and direct carotid artery access along with neuro-protective flow reversal during delivery of the stent. The Minneapolis Heart Institute Foundation (MHIF) was selected as the only center in the four state area to participate in the ROADSTER 2 Study. Jessica Titus, MD, and Timothy Sullivan, MD, vascular surgeons at the Minneapolis Heart Institute at Abbott Northwestern Hospital, will serve as Principal Investigator and Co-Investigator, respectively, for the trial. "We are excited to be able to bring this carotid artery revascularization option to our patients. It combines the advantages of a minimally invasive approach for high risk patients with a lower stroke risk than the standard method for carotid stent placement." said Dr. Titus.
The ENROUTE Transcarotid Neuroprotection System utilizes robust flow reversal during transcarotid stent placement to remove micro and macro emboli throughout the intervention. Erica Rogers, CEO of Silk Road Medical notes, "We selected the staff and researchers at MHIF as a key trial partner because they have the vascular surgery and research expertise we need to participate in this important study and provide this less invasive and more efficient TCAR procedure for their patients presenting with carotid artery disease." The first registry trial participant at MHIF is scheduled for first quarter 2016. In total, the ROADSTER2 study will assess the real world treatment of individuals at risk for stroke due to carotid artery disease with the ENROUTE Transcarotid Neuroprotection (NPS) and Stent System with a minimum of 600 participants around the country.