Briviact approved for epileptic seizures
(HealthDay)—Briviact (brivaracetam) has been approved by the U.S. Food and Drug Administration to treat partial onset seizures in people aged 16 and older with epilepsy.
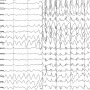
Partial onset seizures describe those that originate in a limited area of the brain, the FDA said Friday in a news release.
Epilepsy, a common disorder believed to affect more than 5 million people in the United States, is characterized by recurring seizures, with typical symptoms including uncontrolled movement, muscle spasms and abnormal behavior.
Common causes of epilepsy include stroke, infection, tumors, brain injury and abnormal brain development, the FDA said. In many cases, no cause can be identified, the agency added.
Briviact, taken with other medications, was evaluated in clinical studies involving more than 1,500 people. Common side effects included drowsiness, dizziness and nausea.
As with other anti-epilepsy medications, Briviact may carry more serious risks including suicidal thoughts or actions, feelings of agitation, new or worsening depression, aggression and panic attacks, the FDA said.
Rarely, users may develop allergic-like reactions that include symptoms such as swelling of the lips, eyelids or tongue, and difficulty breathing, the agency added.
Briviact is marketed by Smyrna, Ga.-based UCB Inc.
More information: Visit the FDA to learn more.
Copyright © 2016 HealthDay. All rights reserved.