Scientists reveal why people with the ApoE4 gene are more susceptible to Alzheimer's disease
For decades, scientists have known that people with two copies of a gene called apolipoprotein E4 (ApoE4) are much more likely to have Alzheimer's disease at age 65 than the rest of the population. Now, researchers at the Salk Institute have identified a connection between ApoE4 and protein build-up associated with Alzheimer's that provides a possible biochemical explanation for how extra ApoE4 causes the disease.
"The big picture here is that we've found a very different way of thinking about how the proteins in Alzheimer's disease might be regulated," says Alan Saghatelian, Salk professor and holder of the Dr. Frederik Paulsen Chair in Salk's Clayton Foundation Laboratories for Peptide Biology. Their findings, which appear in the August 2016 issue of the Journal of the American Chemical Society underscore the importance of looking at genes and proteins not classically associated with Alzheimer's to make progress in understanding the disease.
Late-onset Alzheimer's disease—the subset of the disorder occurring in people age 65 and over—affects more than 5 million Americans, and is characterized by progressive memory loss and dementia. Scientists have put forth a variety of hypotheses on its causes, including the accumulation of protein clusters called beta-amyloid plaques and tau tangles in the brain.
Apolipoprotein E comes in three versions, or variants, called ApoE2, ApoE3 and ApoE4. All the ApoE proteins have the same normal function: carrying fats, cholesterols and vitamins throughout the body, including into the brain. While ApoE2 is protective and ApoE3 appears to have no effect, a mutation in ApoE4 is a well-established genetic risk factor for late-onset Alzheimer's disease. Previous reports have suggested that ApoE4 may affect how the brain clears out beta-amyloid, but what was happening at the molecular level wasn't clear.
"ApoE4 is the most predictive genetic change for late onset Alzheimer's, but no one has really understood what's going on at the molecular level," says Saghatelian. Scientists had previously uncovered hints, however, that ApoE4 might degrade differently than the other variants, but the protein that carried out this breakdown of ApoE4 was unknown.

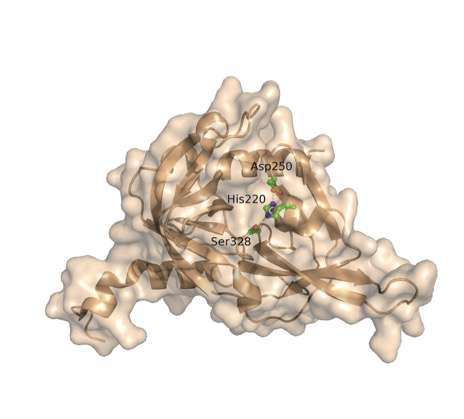
To find the protein responsible for degrading ApoE4, Saghatelian and research associate Qian Chu, first author of the new paper, screened tissues for potential suspects and homed in on one enzyme called high-temperature requirement serine peptidase A1 (HtrA1).
When they compared how HtrA1 degraded ApoE4 with ApoE3, they found that the enzyme processed more ApoE4 than ApoE3, chewing ApoE4 into smaller, less stable fragments. The researchers confirmed the observation in both isolated proteins and human cells. The finding suggests that people with ApoE4 could have less ApoE overall in their brain cells—and more of the breakdown products of the protein.
"There's been an idea tossed around that ApoE4 breakdown products could be toxic," said Saghatelian. "Now, knowing the enzyme that breaks it down, we have a way to actually test this idea."
But it's not just a lack of full-length ApoE or an increase in its fragments that may be causing Alzheimer's in people with ApoE4. Saghatelian and Chu also found that ApoE4—because it binds so well to HtrA1—keeps the enzyme from breaking down the tau protein, responsible for tau tangles associated with Alzheimer's.
"People have thought about tau or amyloid beta as things that cause Alzheimer's, but this suggests that we need to think more globally about proteins that could be impacting tau or amyloid through biochemical pathways," Saghatelian said.
The results need be tested and confirmed in animal studies before researchers can be sure that HtrA1 is the link between ApoE4 and Alzheimer's in humans. But if they hold true, they could point toward a better understanding of the disease and potential new treatment strategies.
More information: Qian Chu et al. HtrA1 Proteolysis of ApoE In Vitro Is Allele Selective, Journal of the American Chemical Society (2016). DOI: 10.1021/jacs.6b03463