How AIDS conquered North America: Researchers restore HIV genomes from serum samples more than 40 years old
Researchers at the University of Arizona and the University of Cambridge in the U.K. have reconstructed the origins of the AIDS pandemic in unprecedented detail.
The findings were made possible by a molecular technique the team developed for this project, enabling them to recover genetic material from more than 40-year-old serum samples and decipher the gene sequence of the human immunodeficiency virus, or HIV, subtype that started the outbreak on the North American continent in the early 1970s. Phylogenetic analyses estimate the jump to the U.S. at about 1970 and place the ancestral U.S. virus in New York City, strongly suggesting this was the crucial hub from which HIV made its way across the continent.
Insights gained from this study may help researchers and health officials better understand how pathogens move through populations and lead to more effective strategies aimed at reining in, or eradicating, dangerous pathogens.
The results will be published in the advance online publication of Nature on Oct. 26. They confirm previous findings retracing the routes by which the virus entered and spread through the U.S. and eliminate any remaining doubt surrounding the Caribbean region as a key steppingstone from which HIV jumped into the U.S. The paper also reports the first recovery of the full HIV-1 genome from an individual known as "Patient Zero" and shows that there is neither biological nor historical evidence for the widely held belief that he was the primary cause of the HIV epidemic in North America.
While it had been established that HIV already was infecting people in the U.S. before 1981, the year AIDS was recognized, the timing and earliest movements of the virus in the U.S. were unknown until now. Leading an interdisciplinary team of scientists, Michael Worobey, an expert on virus evolution, and Richard McKay, a scholar specializing in the history of public health, set out on a quest to unravel the secrets surrounding the AIDS epidemic as it unfolded. The endeavor called for new molecular techniques that would make it possible to recover and "restore" genetic material from samples whose age and condition made them intractable to existing analytic methods.
"Standard methodology such as antibody-detecting serological blood tests will tell you whether a person had HIV, but you might not be able to get any of the HIV gene sequences out of it, because to do that, you need the RNA from the virus," says Worobey, a professor and head of the UA's Department of Ecology and Evolutionary Biology. "The virus' RNA is an extremely delicate molecule comprising 10,000 nucleotides, and breaks down very quickly."
Worobey's lab developed a technique called RNA jackhammering, which breaks down the huge human genome in the samples into tiny overlapping chunks and extracts the RNA of the virus.
"We then very carefully amplify the RNA of the virus without letting the background RNA get in the way," he explains.
Worobey says the technique may hold potential for other health care applications, such as more sensitive bioassays for screening blood samples for cancer markers or viruses, including Zika.
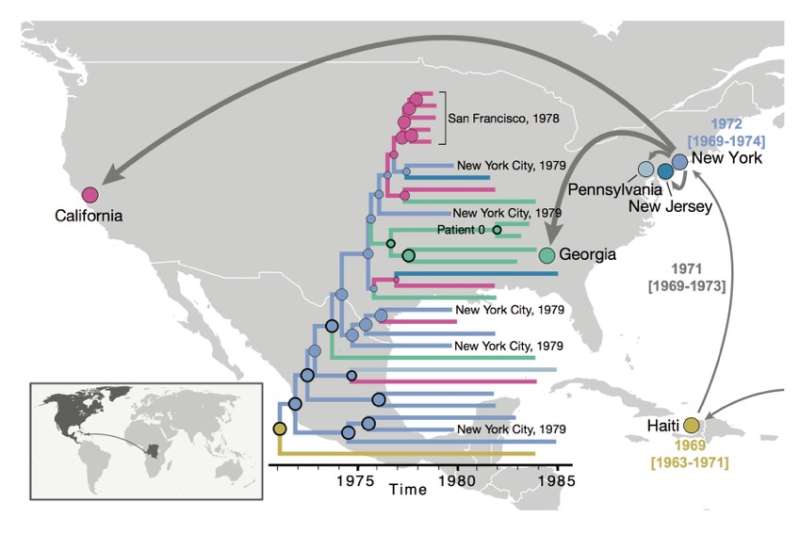
By screening more than 2,000 serum samples collected from U.S. men between 1978 and 1979, all of which degraded over time, the technique allowed the researchers to recover eight near full-length viral RNA genome sequences, representing the oldest HIV genomes in North America. This early, full-genome "snapshot" reveals the U.S. HIV-1 epidemic showed surprisingly extensive genetic diversity in the 1970s but also provides strong evidence of its emergence from a pre-exiting Caribbean epidemic.
Having the complete genomic information in front of them allowed the authors to tackle questions that had vexed researchers—for example, how quickly the virus was spreading at different times in different locations. Once HIV had crossed the Atlantic from Africa, it quickly spread through the Caribbean and from there into the U.S. Yet, the epidemic went unnoticed until it hit the U.S. New York City turned out to be the most critical hub for the AIDS epidemic in the U.S., and the newly sequenced genomes showed the virus must have jumped there in, or very near, 1970. From there, HIV spread to San Francisco and presumably to other locations in California, where AIDS patients were first recognized in 1981.
"In New York City, the virus encountered a population that was like dry tinder," Worobey explains, "causing the epidemic to burn hotter and faster and infecting enough people that it grabs the world's attention for the first time.
"That information is stamped into the RNA of the virus from 1970," he says. "Our analysis shows that the outbreaks in California that first caused people to ring the alarm bells and led to the discovery of AIDS were really just offshoots of the earlier outbreak that we see in New York City."
From the genetic data, the team was able to construct evolutionary trees of the various HIV strains and how they spread through the U.S. They revealed that by the late 1970s HIV had diversified in almost the same genetic diversity we see today.
"Right around 1970, we see the signal of emergence of this virus, which is evidence that it had to have been there at that point in time in at least one person," Worobey says.
Being able to look back in time and piece together what it took for the HIV pandemic to happen is encouraging, the researchers say.
"Now we can now look forward in time and really see a future in which—even if the virus is not completely eliminated—it could be driven down to no new transmission in large swaths of the world," Worobey says.
The molecular biology assays developed in this work could lead to more sensitive tests that detect the virus sooner in people who are unaware that they were infected very recently, he says.
"Earlier detection and better alignment of the various options we have to make it harder for the virus to move from one person to the next," he says, "are key to driving HIV out of business."
More information: Michael Worobey et al, 1970s and 'Patient 0' HIV-1 genomes illuminate early HIV/AIDS history in North America, Nature (2016). DOI: 10.1038/nature19827