October 28, 2016 report
Study shows how newborn neurons associated with new experiences are integrated into existing networks
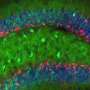
(Medical Xpress)—A team of researchers with the Fundación Instituto Leloir in Argentina has found that newborn neurons in the brains of mice become more complex if they are exposed to richer experiences than do those in the brains of mice kept in simple cages. In their paper published in the journal Science, the team describes experiments they conducted on mouse brains and what they learned about the development of dentate gyrus neurons.
Prior research has shown that most cells in mammalian brains do not divide, which means that new cells do not come about (which is why brain damage is usually permanent)—but there are some, particularly in the hippocampus, that do—these are called dentate gyrus neurons or granule cells; scientists believe they are part of the process that involves memory creation and retention. In this new effort, the researchers sought to learn more about what occurs as they develop.
To trace the development process, the researchers introduced a fluorescent protein to the cells to cause them to turn red. They allowed some of the mice to roam around in an enriched environment while others were confined to their cages for a three-week period—the granule cells take six weeks to fully mature. Then they assessed the developing cells to see if there were any differences between the two groups. They report that the granule cells in mice that were allowed to traverse tunnels and play with objects had longer dendrites and an increase in connections with other neurons than did new neurons in those mice kept in simple cages.
The team then conducted additional optogenetic and chemical experiments on the developing cells and found that surrounding mature cells played a role in activating newly created cells via intermediaries called interneurons. They also found that they could recreate the process artificially and that blocking such stimulation slowed the development of longer dendrites in mice raised in the enriched environment.
The researchers note that there are still many questions to be answered about the process, such as whether the enriched environment was enhancing the brain, thereby making the mice better learners improving their memory, or if those unexposed to a rich environment were the norm, rather than stunted.
More information: D. D. Alvarez et al. A disynaptic feedback network activated by experience promotes the integration of new granule cells, Science (2016). DOI: 10.1126/science.aaf2156
Abstract
Experience shapes the development and connectivity of adult-born granule cells (GCs) through mechanisms that are poorly understood. We examined the remodeling of dentate gyrus microcircuits in mice in an enriched environment (EE). Short exposure to EE during early development of new GCs accelerated their functional integration. This effect was mimicked by in vivo chemogenetic activation of a limited population of mature GCs. Slice recordings showed that mature GCs recruit parvalbumin γ-aminobutyric acid–releasing interneurons (PV-INs) that feed back onto developing GCs. Accordingly, chemogenetic stimulation of PV-INs or direct depolarization of developing GCs accelerated GC integration, whereas inactivation of PV-INs prevented the effects of EE. Our results reveal a mechanism for dynamic remodeling in which experience activates dentate networks that "prime" young GCs through a disynaptic feedback loop mediated by PV-INs.
© 2016 Medical Xpress