To treat one rare blood disorder, scientists exploit another
For the nearly 400,000 individuals around the world with hemophilia A and hemophilia B—rare blood disorders that impair a person's ability to form clots to stop bleeding—relief may someday come from a treatment with similarities to another blood disorder, known as factor V Leiden.
In a study published online today in Blood, the Journal of the American Society of Hematology (ASH), researchers report that they have developed a new therapy that gives the clotting process more time to produce thrombin, an enzyme needed for clotting. They suggest this treatment could someday help all patients with hemophilia, including those who develop antibodies against standard therapy.
Hemophilia is caused by a deficiency in certain clotting factors, factor VIII for hemophila A and factor IX for hemophilia B. Standard treatment for hemophilia is administration of the missing factor. However, this requires every-other-day intravenous injections, is under-effective, and in some cases results in the development of antibodies that reject the factor, so researchers tried a new approach based on observations relating to a common mutation associated with excessive clotting, factor V Leiden.
"We know that patients who have severe hemophilia and also have mutations that increase clotting, such as factor V Leiden, experience less-severe bleeding," said study co-author Trevor Baglin, MD, of Cambridge University's Addenbrooke's Hospital.
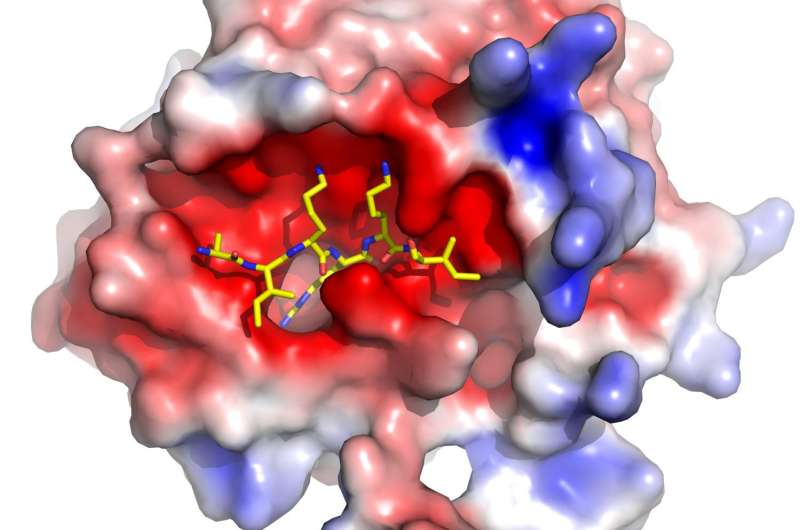
Dr. Baglin and colleagues pursued a novel strategy to reduce the activity of an enzyme—activated protein C (APC)—that promotes bleeding. In patients with factor V Leiden, defects in the anticoagulant APC mechanism lead to an overactive production of thrombin, an enzyme that is needed to form a clot. The team, led by Professor James A. Huntington, PhD, exploited this insight by developing a direct inhibitor of APC. Researchers modified serine protease inhibitors, known as serpins, to make them specific and efficient inhibitors of APC.
"We hypothesized that if we targeted the protein C pathway we could prolong thrombin production," said Dr. Huntington, of the Cambridge Institute for Medical Research at the University of Cambridge. "We engineered a serpin so that it could selectively prevent APC from shutting down thrombin production before the formation of a stable clot."
To test their theory, the team administered the serpin to mice with hemophilia B and clipped their tails. In this model, the blood loss decreased as the dose increased, with the highest dose reducing bleeding to the level of the healthy mice. Further injury models underscored that the serpin helped the majority of the mice form stable clots, with higher doses resulting in quicker clot formation. The serpin was also able to accelerate clot formation when added to blood samples from hemophilia A patients.
"It is our understanding that because we are targeting a general anti-clotting process, our serpin could effectively treat patients with either hemophilia A or B, including those who develop inhibitors to more traditional therapy," stated Dr. Huntington. He went on to add, "Additionally, we have focused on engineering the serpin to be both subcutaneously delivered and long-acting. This will free patients from the cumbersome thrice-weekly infusions that are necessary under many contemporary therapy regimens."
Explaining the roadmap for this therapy's further development, Dr. Baglin noted, "Within three years, we hope to be conducting our first-in-man trials of a subcutaneously-administered form of our serpin. It is important to remember that the majority of people in the world with hemophilia have no access to therapy. A stable, subcutaneous, long-acting, effective hemostatic agent could bring treatment to a great deal many more hemophilia sufferers."
More information: Design and characterization of an APC-specific serpin for the treatment of hemophilia, Blood 2016 :blood-2016-05-718635; DOI: 10.1182/blood-2016-05-718635 , www.bloodjournal.org/content/e … blood-2016-05-718635