Trial determines safest airway device for babies in surgery
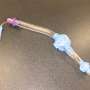
A ground-breaking Western Australian medical trial, involving babies with breathing problems, has determined that a laryngeal mask (a medical device that keeps a patient's airway open) is the preferred option over an endotracheal tube during minor surgical procedures.
A team of researchers from The University of Western Australia, Princess Margaret Hospital (PMH), Telethon Kids Institute and Curtin University performed a randomised clinical trial on infants (aged less than one year) to improve the safety of anaesthesia for babies.
The research, which was funded by Perth Children's Hospital Foundation donors and NHMRC (via UWA), was led by Professor Britta von Ungern-Sternberg, PMH and The University of Western Australia Chair of Paediatrics, and by Associate Professor Graham Hall, Head of the Children's Lung Health team at Telethon Kids Institute.
The trial, which was completed at the Department of Anaesthesia and Pain Management at Princess Margaret Hospital in Perth, compared two airway devices commonly used during anaesthesia and evaluated their effect on the incidence of infants suffering from breathing problems during or shortly after the procedure.
Breathing problems occur in many babies undergoing anaesthesia. Although most breathing problems do not cause long lasting harm, in some very rare cases these problems can lead to significant complications.
During the trial, 181 babies were randomly assigned to receive either a laryngeal mask or an endotracheal tube during surgery. Infants receiving an endotracheal tube were found to be three times more likely to develop breathing problems compared with infants that received the less invasive laryngeal mask airway. The difference was even larger when looking at more serious breathing problems where the difference was fivefold.
The trial results show that the use of a laryngeal mask results in significantly fewer incidents of respiratory problems and should be considered amongst other patient and surgery related factors when selecting an airway device for a specific patient.
Professor Britta von Ungern-Sternberg said the trial was a major step towards improving the safety of infants undergoing anaesthesia for minor elective surgery.
"However, we have to be mindful that the laryngeal mask airway will not suit all infants undergoing all types of surgery," she said.
Associate Professor Graham Hall said the study provided evidence for paediatric anaesthetists on ways they could further improve the safety of surgery on babies.
"The next step will be to perform trials to establish to what extent the results can be generalised across paediatric settings worldwide," Associate Professor Hall said.