How depression can muddle thinking
Depression is associated with sadness, fatigue and a lack of motivation. But people with depression can also have trouble processing information and solving problems. Now scientists studying a rat model for depression are identifying on a molecular level how the condition could affect thinking. The findings, published in the journal ACS Chemical Neuroscience, could lead to the development of new depression treatments that would address associated cognitive problems.
The World Health Organization estimates that globally, 350 million people of all ages suffer from depression. Among the mix of symptoms that characterize the condition are cognitive dysfunctions—difficulty concentrating, remembering details and making decisions. These symptoms tend to linger even as other symptoms fade, which can seriously hamper a person's ability to work or study. Marco A. Riva and colleagues wanted to determine how depression might affect the brain on a molecular level.
The researchers found that certain proteins were expressed in higher amounts in a control group of rats in response to a standard cognitive test—novel object recognition—than in rats with symptoms characteristic of human depression. Other recent studies have suggested that these proteins, including oligophrenin-1 and Bmal1, play a role in cognitive processes. The researchers say that their findings could help inform future searches for treatments for major depression and stress-related disorders.
More information: Francesca Calabrese et al. Chronic Mild Stress-Induced Alterations of Local Protein Synthesis: A Role for Cognitive Impairment, ACS Chemical Neuroscience (2017). DOI: 10.1021/acschemneuro.6b00392
Abstract
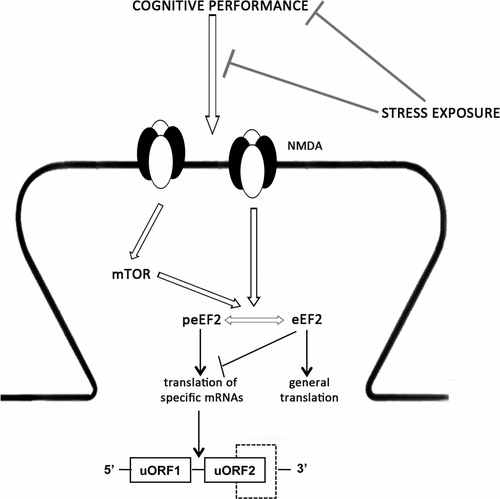
Depression, a major cause of disability worldwide, is characterized by a complex and heterogeneous symptomatology. With this respect, cognitive deterioration represents a major problem that has a strong impact on a patient's function. Thus, within the context of a depressive phenotype, it is important to characterize the mechanisms that sustain cognitive dysfunctions and may represent an important target for pharmacological intervention. Here, using the chronic mild stress (CMS) paradigm of depression, we found that, independently from the anhedonic phenotype, CMS rats showed a deficit in the novel object recognition (NOR) test, which is associated with an inability to phosphorylate GluN2B subunit on Ser1303 and to activate the mTOR pathway. In agreement with the role of these systems in the control of local protein synthesis, we observed an increase phosphorylated eukaryotic elongation factor 2 (eEF2) in the crude synaptosomal fraction after the NOR test specifically in control animals. Since it has been demonstrated that peEF2 leads to the translation of specific mRNAs, we investigated if the gene-specific translational control depends on the presence of upstream open reading frame (uORF). Interestingly, we found a significant increase of oligophrenin-1 (2 uORFs) and of Bmal1 (7 uORFs) protein levels specifically in the control animals exposed to the NOR test. Our results demonstrated that the cognitive decline associated with stress exposure might be due to alterations in local protein translation of specific mRNAs, suggesting that a pharmacological intervention able to correct these defects might be useful in the improvement of deteriorated functions in patients with major depression and stress-related disorders.