First-void morning urine not necessary for CIN2+ detection
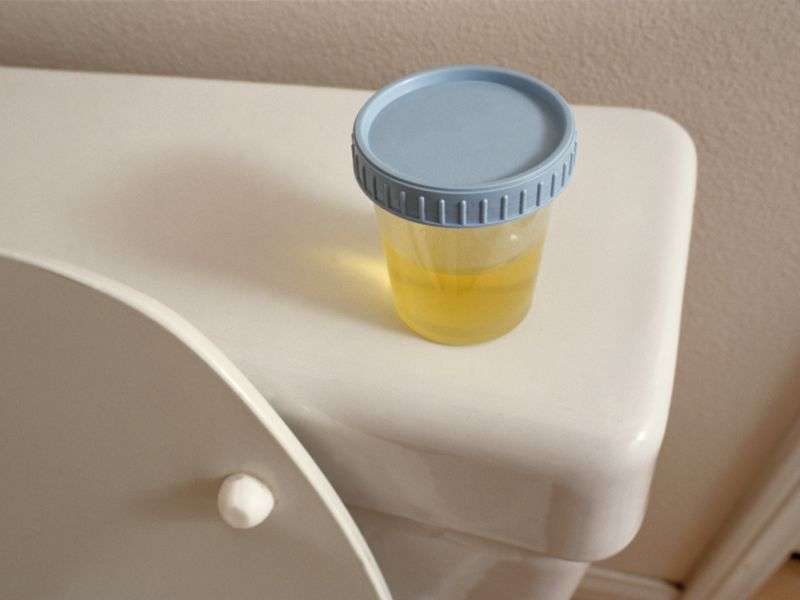
(HealthDay)—There is no advantage in testing morning first-void urine over later samples for cervical intraepithelial neoplasia grade 2+ (CIN2+) detection using human papillomavirus (HPV) testing, according to a study published online April 9 in BJOG: An International Journal of Obstetrics and Gynaecology.
Annemiek Leeman, from DDL Diagnostic Laboratory in Rijswijk, Netherlands, and colleagues sent women undergoing colposcopy with biopsy for abnormal Pap smears a device to self-collect urine samples on the morning of colposcopy (U1) and later in the day (U2). Self-collected urine samples were compared to brush-based self-samples (SS) and physician-taken smears (PTS) for ability to detect CIN2+. Samples were tested for HPV DNA using two assays (the analytically sensitive SPF10-DEIA-LiPA25 assay and the clinically validated GP5+/6+-EIA-LMNX).
The researchers found, based on samples from 91 patients analyzed, that all CIN3 cases (six patients) were high-risk HPV (hrHPV)-positive in PTS, SS, U1, and U2 with both HPV assays. Using the SPF10 assay sensitivity for CIN2+ was 100, 100, 95, and 100 percent, respectively, versus 95 percent in all sample types with the GP5+/6+ assay. The differences in sensitivities and specificities for both tests on each of the sample types were not significant. The discordance on hrHPV genotype was 10 to 14 percent.
"CIN2+ detection using HPV testing in first-void urine shows sensitivity similar to that of physician-taken smears or brush-based self-samples and is convenient," the authors write. "There was substantial to almost excellent agreement between all samples on genotype with both hrHPV assays. There was no advantage in testing morning first-void urine over later samples."
More information:
Abstract
Full Text (subscription or payment may be required)
Editorial (subscription or payment may be required)
Copyright © 2017 HealthDay. All rights reserved.