New interferon shows promise against hepatitis B in cell culture, and animal model
Hepatitis B is notoriously difficult to eradicate with currently available agents.. Now, in a new study, a novel form of "pegylated" interferon-β has reduced hepatitis B virus (HBV) infections in experimental human-derived cells and in mice more effectively than the conventional pegylated interferon-α2a, suggesting that it could lead to improved treatment for hepatitis B infection in humans. The research is published in Antimicrobial Agents and Chemotherapy, a journal of the American Society for Microbiology.
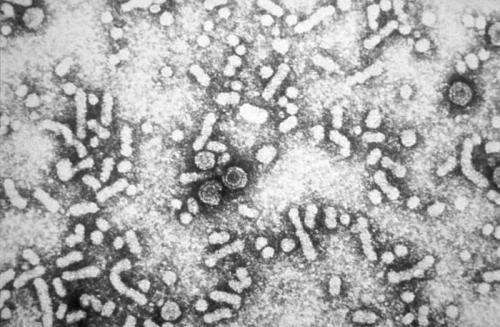
In the study, a team of Japanese investigators compared the antiviral potency of pegylated interferon-α2a with that of their novel agent, known by the alpha-numeric TRK-560, on certain experimental lines of HBV-infected human liver cancer cells, and in mouse models inoculated with HBV-containing serum, and with livers modified to be largely composed of human liver cells. TRK-560's potency significantly exceeded that of the conventional agent. (image: electron micrograph of HBV, Wikimedia)
Several problems had plagued pegylated interferon-α2a and conventional interferon-β. First, in both cases, repeated exposure leads to reduced sensitivity, resulting in decreasing effectiveness of this compound. Second, both compounds trigger immune responses in which antibodies neutralize these compounds, rendering them ineffective.
Additionally, interferons are vulnerable to enzymes that cleave proteins, which are quite normally generated from cells all over the body. These enzymes aren't necessarily directed at interferons, but when they come into contact with them—through random movement—they slice the interferons, eliminating their activity.
Pegylation—the attachment of polyethylene glycol (PEG) to the interferon-reduces the induction of neutralizing antibodies, and also helps prevent protein cutting enzymes from cleaving them. Part of TRK-560's advantage over pegylated alpha interferon stems from the fact that the attachment of PEG to the latter is somewhat random, whereas in TRK-560, PEG is attached only at an optimal location within the compound.
And indeed, TRK-560 was more effective than pegylated interferon-α2a and interferon-β.
The investigators also showed that TRK-560 more strongly induced expression of genes that are stimulated by interferon. Several hundred such genes exist, and have antiviral and other immune-related functions, and they suppress viral replication, and degrade viral proteins, said Masataka Tsuge, MD, PhD, assistant professor of Natural Science, Center for basic Research and Development, Hiroshima University, Japan. They share a common regulatory element that causes them all to be upregulated in response to interferon, he said.
"In effect, the cell already has a potent defense against HBV, but the virus tries to prevent its activation," explained Tsuge. "The goal of interferon therapy is to overcome this interference and trigger the activation of this pathway."
"Despite improvements in the antiviral treatment of chronic hepatitis B, it is difficult to eliminate the HBV genome, which remains resident inside the nuclei of human hepatocytes in the form of "covalently closed, circular" DNA," said Tsuge. "Therefore, it is very important to develop novel drugs that can eliminate or permanently suppress HBV genomes encamped within human hepatocytes."
Interestingly, TRK-560 might also have potential as a novel therapeutic agent for malignancies, according to work by several coauthors on the current paper, said Tsuge. "Interferon potently activates the innate immune system, which not only acts to suppress viral infection, but is also essential in combating malignancies." "Innate immunity" refers to nonspecific defense mechanisms that come into play soon after an antigen appears in the body. These can include chemicals that circulate in the blood, such as interferons, and immune cells that attack and engulf foreign invaders.