Blocking yeast-bacteria interaction may prevent severe biofilms that cause childhood tooth decay
Though most tooth decay can be blamed on bacteria, such as Streptococcus mutans, the fungus Candida albicans may be a joint culprit in an alarmingly common form of severe tooth decay affecting toddlers known as early childhood caries.
In earlier research, a team from the University of Pennsylvania School of Dental Medicine had found that C. albicans, a type of yeast, took advantage of an enzyme produced by S. mutans to form a particularly intractable biofilm. In a new study, the researchers have pinpointed the surface molecules on the fungus that interact with the bacterially-derived protein. Blocking that interaction impaired the ability of yeast to form a biofilm with S. mutans on the tooth surface, pointing to a novel therapeutic strategy.
"Instead of just targeting bacteria to treat early childhood caries, we may also want to target the fungi," said Hyun (Michel) Koo, senior author on the study and a professor in the Department of Orthodontics and Divisions of Pediatric Dentistry and Community Oral Health. "Our data provide hints that you might not need to use a broad spectrum antimicrobial and might be able to target the enzyme or cell wall of the fungi to disrupt the plaque biofilm formation."
Koo collaborated on the work with Penn Dental's Geelsu Hwang, the first author and a research assistant professor, as well as Yuan Liu, Dongyeop Kim and Yong Li. Damian J. Krysan of the University of Rochester was also a coauthor.
The research appears in the journal PLOS Pathogens.
Candida can't effectively form plaque biofilms on teeth on its own nor can it bind S. mutans, unless in the presence of sugar. Young children who consume sugary beverages and foods in excess are at risk of developing early childhood caries. Koo's team had previously discovered that an enzyme, GftB, secreted by S. mutans, uses sugar from the diet to manufacture glue-like polymers called glucans. Candida promotes this process, resulting in a sticky biofilm that allows the yeast to adhere to teeth and to bind to S. mutans.
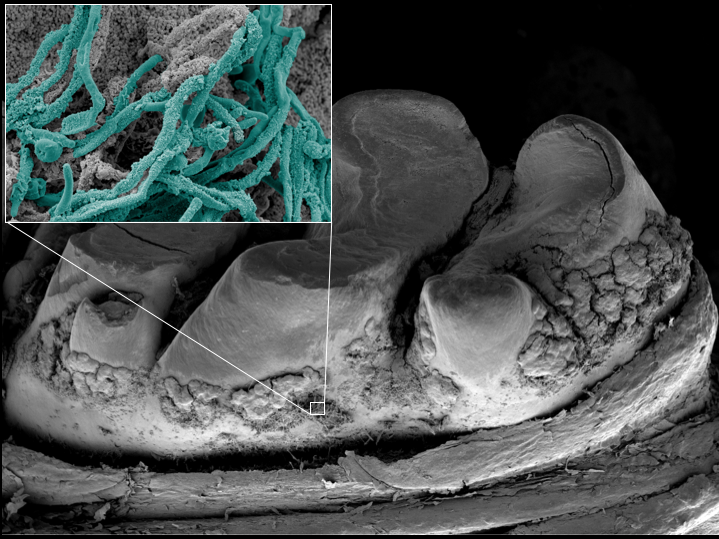
The researchers suspected that the outer portion of the Candida cell wall, composed of molecules called mannans, might be involved in binding GftB. To gain a more detailed understanding of the interaction between the yeast and the enzyme, the researchers measured the binding strength between various mutant Candida strains and GtfB using biophysical methods. Such measurements were developed by Hwang, who has a background in engineering and is applying his unique expertise to advance dental science.
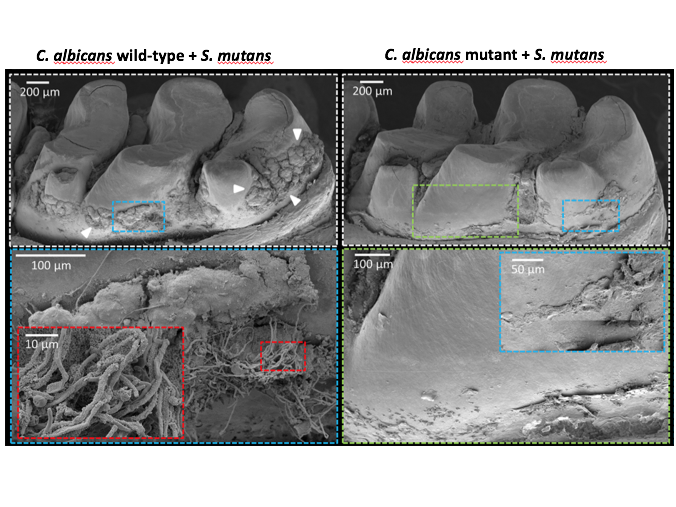
Koo, Hwang and colleagues found that the enzyme bound much more weakly to mutants that lacked components of the mannan layer than the wild-type Candida. The team next looked at the abilities of the mutant Candida to form biofilms with S. mutans in a laboratory assay. The mutants that had impaired binding with GftB were mostly unable to form biofilms with S. mutans, resulting in significantly fewer Candida cells and reduced production of the sticky glucans molecules.
Using another biophysical technique, the researchers tested how stable the biofilms were when attached to a tooth-like surface. While low-shear stress, roughly equivalent to the force generated by taking a drink of water, removed only a quarter of the wild-type biofilm, the same force removed 70 percent of the biofilms with mutant Candida. When the forces were increased to the equivalent of a vigorous mouth rinse, the mutant biofilms were almost completely dislodged.
To ensure their findings translated to in vivo conditions, they examined biofilm formation in a rodent model that can mimic the development of early childhood caries. When animals were infected with both S. mutans and either the wild type or defective mutant yeast strains, the researchers observed clear differences. While biofilm formation was abundant if the wild-type yeast was used, it was substantially reduced in animals infected with the mutant strain. A more precise analysis revealed that the these defective biofilms completely lacked viable Candida cells and S. mutans were reduced by more than five-fold.
The findings point to a new direction for treatment of early childhood caries. The current standard of care, beyond the use of fluoride as a preventive approach, is to target only the bacteria with antimicrobials, or to use surgical interventions if the tooth decay has become too severe.
"This disease affects 23 percent of children in the United States and even more worldwide," said Koo. "In addition to fluoride, we desperately need an agent that can target the disease-causing biofilms and in this case not only the bacterial component but also the Candida."
Koo and colleagues are now working on novel therapeutic approaches for targeted interventions, which can be potentially developed for clinical use.
More information: Geelsu Hwang et al, Candida albicans mannans mediate Streptococcus mutans exoenzyme GtfB binding to modulate cross-kingdom biofilm development in vivo, PLOS Pathogens (2017). DOI: 10.1371/journal.ppat.1006407