New mutations related to hereditary neuroendocrine tumours
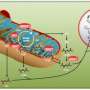
Pheochromocytomas and paragangliomas are rare neuroendocrine tumours with a strong hereditary component. Half the genes whose alterations confer hereditary susceptibility to this condition code for enzymes involved in the Krebs cycle, a metabolic route involved in cellular respiration. A study by the Hereditary Endocrine Cancer Group of the Spanish National Cancer Research Centre (CNIO), and published in the journal Clinical Cancer Research, identifies new genes associated with this cycle that are involved in the development of these tumours.
Approximately 40 percent of patients diagnosed with pheochromocytomas or paragangliomas—considered to be the same condition but affecting different parts of the body—harbour a germline alteration in susceptibility genes related to these diseases. This feature earns them the dubious honour of being the tumours with the strongest hereditary component known to date.
Over the past few years the Hereditary Endocrine Cancer Group has focused its efforts on discovering new genes with a hereditary susceptibility to developing this condition employing the very latest sequencing technologies to do so. In addition to the 2011 discovery of germline mutations in MAX in patients with hereditary pheochromocytoma, a new gene implicated in the Krebs cycle (MDH2) was identified in 2015 as responsible for a hereditary susceptibility to pheochromocytomas and paragangliomas. The mutations in MDH2, as well as in other genes implicated in the Krebs cycle (such as FH and the SDH genes) cause an accumulation of oncometabolites that inhibit the activity of various enzymes implicated in DNA demethylation, which leads to hypermethylation of certain regions of the DNA—CpG islands—and ultimately, alterations in gene expression.
In the study published in the current issue of Clinical Cancer Research, the researchers selected tumour samples that showed this characteristic pattern of hypermethylation but did not have any of the known mutations. Thanks to massive sequencing of all of the genes implicated in the Krebs cycle and to the study of the cycle's intermediaries and of the methylation profile of the tumours, the authors have identified several susceptibility genes associated with a risk of developing this pathology.
In the study, the authors show that the presence of a germline mutation in the GOT2 gene, which was found in a patient with nine metastatic tumours, gives rise to increased activity of the encoder enzyme. Furthermore, the authors describe extraordinarily rare mutations in two patients: one epimutation in the SDHC gene and another in the IDH1—the third to be identified in this gene. Finally, in an extension study focused on more than 60 patients with these neuroendocrine tumours, a new susceptibility gene, IDH3B, was associated with the development of paragangliomas.
"The identification of mutations in the Krebs cycle genes is especially important, given that the patients diagnosed with them have a greater likelihood of developing metastasis," explains Alberto Cascón, from the Hereditary Genetic Cancer Group and leader of the study. "The Krebs cycle could be the Achilles heel of these tumours, and therefore, a potential target for future therapies," adds Mercedes Robledo, head of the group and member of the team that carried out the research.
More information: Laura Remacha et al, Targeted exome sequencing of Krebs cycle genes reveals candidate cancer predisposing mutations in pheochromocytomas and paragangliomas, Clinical Cancer Research (2017). DOI: 10.1158/1078-0432.CCR-16-2250