New cardiac catheter combines light and ultrasound to measure plaques

To win the battle against heart disease, cardiologists need better ways to identify the composition of plaque most likely to rupture and cause a heart attack. Angiography allows them to examine blood vessels for constricted regions by injecting them with a contrast agent before X-raying them. But because plaque does not always result in constricted vessels, angiography can miss dangerous buildups of plaque. Intravascular ultrasound can penetrate the buildup to identify depth, but lacks the ability to identify some of the finer details about risk of plaque rupture.
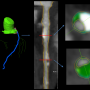
Professor Laura Marcu's lab in the Department of Biomedical Engineering at UC Davis has now combined intravascular ultrasound with fluorescence lifetime imaging (FLIm) in a single catheter probe that can image the tiny arteries of a living heart. The new catheter can simultaneously retrieve structural and biochemical information about arterial plaque that could more reliably predict heart attacks.
The new device is described in a recent paper published in Scientific Reports.
An optical fiber in the catheter sends short laser pulses into surrounding tissue, which fluoresces with tiny flashes of light in return. Different kinds of tissue (collagen, proteins, lipids) emit different amounts of fluorescence.
At the same time, an ultrasound probe in the catheter records structural information about the blood vessel.
Seeking FDA Approval for Human Trials
The combination FLIm-IVUS imaging catheter provides a comprehensive insight into how atherosclerotic plaque forms, aiding diagnosis and providing a way to measure how plaques shrink in response to therapy.
The new catheter has been tested in living swine hearts and samples of human coronary arteries.
The catheter used in the study is flexible enough to access coronary arteries in a living human following standard procedures. It does not require any injected fluorescent tracers or any special modification of the catheterization procedures.
The new technique could not only can improve understanding of mechanisms behind plaque rupture - an event with fatal consequences- but also the diagnosis and treatment of patients with heart disease.
Marcu's group is currently working to obtain FDA approval to test this new intravascular technology on human patients.
More information: Julien Bec et al, In vivo label-free structural and biochemical imaging of coronary arteries using an integrated ultrasound and multispectral fluorescence lifetime catheter system, Scientific Reports (2017). DOI: 10.1038/s41598-017-08056-0