DPP-4I not tied to increased risk of acute pancreatitis in seniors
(HealthDay)—For older adults, dipeptidyl peptidase 4 inhibitors (DPP-4Is) are not associated with increased risk of acute pancreatitis, according to a study published online April 4 in Diabetes Care.
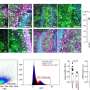
Jin-Liern Hong, Ph.D., from the University of North Carolina at Chapel Hill, and colleagues examined the risks of acute pancreatitis among U.S. Medicare beneficiaries, aged 66+ years, initiating DDP-4Is versus thiazolidinediones or sulfonylureas. Participants had no history of pancreatic disease or alcohol-related diseases.
The researchers found that the risk of acute pancreatitis was not increased comparing 49,374 DPP-4I initiators with 132,223 sulfonylurea initiators (weighted hazard ratio, 1.01; 95 percent confidence interval, 0.83 to 1.24) and comparing 57,301 DPP-4I initiators with 32,612 thiazolidinedione initiators (weighted hazard ratio, 1.11; 95 percent confidence interval, 0.76 to 1.62). The association was not modified by age and sex. Acute pancreatitis incidence was increased for DDP-4I and sulfonylurea initiators but not for thiazolidinedione initiators (2.3 and 2.4 versus 1.5 per 1,000 person-years, respectively), among patients with cardiovascular disease (CVD). The risk of acute pancreatitis was increased among patients with CVD taking DPP-4Is versus thiazolidinediones (weighted hazard ratio, 1.84; 95 percent confidence interval, 1.02 to 3.35), but not compared to sulfonylureas.
"Our study provides evidence that DPP-4I is not associated with an increased risk of acute pancreatitis in older adults overall," the authors write.
Several authors disclosed financial ties to the pharmaceutical industry.
More information: Abstract/Full Text (subscription or payment may be required)
Copyright © 2018 HealthDay. All rights reserved.