August 30, 2018 report
Structure of tau filaments in patients with Pick's disease determined
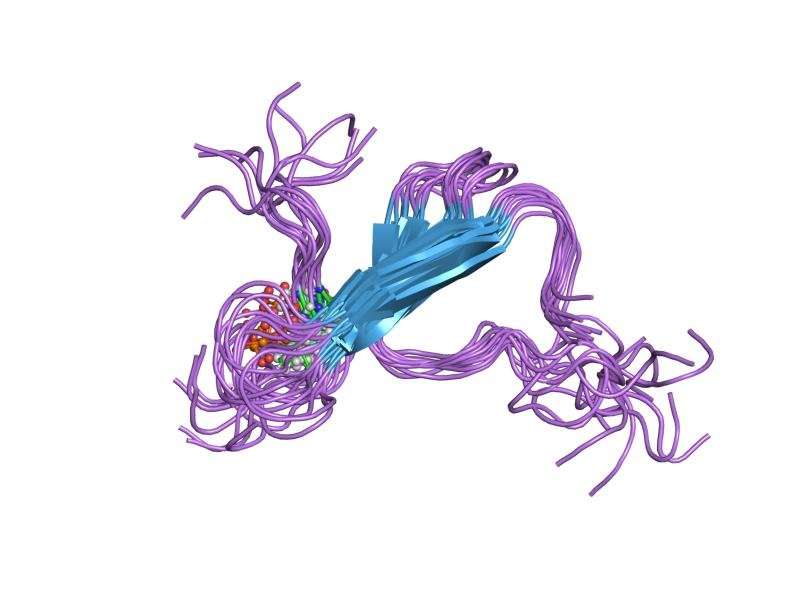
A team of researchers with the MRC Laboratory of Molecular Biology in the U.K. and Indiana University School of Medicine in the U.S. has determined the structure of tau filaments in patients with Pick's disease. In their paper published in the journal Nature, the group describes the technique and technology they used to discover the shape of the folds in the brain and what they found.
Neurological diseases are often characterized by misfolded tau proteins in the brain that lead to the destruction of neurons. Prior research has led to the discovery that there are six tau protein shapes in the human brain, and all of them are essential for normal neuronal activity. For unknown reasons, these proteins sometimes fold improperly, which leads to a cascading effect in which more become misfolded—such cascades are referred to as filaments, and the cascading effect is what leads to degeneration of neurons, and in most patients, death from neurological disease. The researchers with this new effort have been working to determine the structure of the misfolds involved in such diseases, hoping to an understand why proteins misfold, and perhaps find a way to stop it from happening. Recently, they announced that they had determined the structure of tau filaments associated with Alzheimer's disease. In this new effort, they have now done the same with Pick's disease—a degenerative neurological disease that results in destruction of neurons in the frontal lobe.
Tau proteins can have a structure made up of either three or four microtubule-binding repeats, and filaments can have either or both structures. The researchers discovered that such filaments associated with Pick's disease had just three repeats, and that they were novel in shape and distinct from those found in patients with Alzheimer's disease. The technique involved using electron cryomicroscopy, in which samples were cooled to cryogenic temperatures and then examined with an electron microscope. The finding offers evidence to back up a theory that suggests the differences in neurological diseases are likely due to differences in filament structures.
More information: Benjamin Falcon et al. Structures of filaments from Pick's disease reveal a novel tau protein fold, Nature (2018). DOI: 10.1038/s41586-018-0454-y
Abstract
The ordered assembly of tau protein into abnormal filamentous inclusions underlies many human neurodegenerative diseases. Tau assemblies seem to spread through specific neural networks in each disease, with short filaments having the greatest seeding activity. The abundance of tau inclusions strongly correlates with disease symptoms4. Six tau isoforms are expressed in the normal adult human brain—three isoforms with four microtubule-binding repeats each (4R tau) and three isoforms that lack the second repeat (3R tau). In various diseases, tau filaments can be composed of either 3R or 4R tau, or of both. Tau filaments have distinct cellular and neuroanatomical distributions5, with morphological and biochemical differences suggesting that they may be able to adopt disease-specific molecular conformations. Such conformers may give rise to different neuropathological phenotypes, reminiscent of prion strains10. However, the underlying structures are not known. Using electron cryo-microscopy, we recently reported the structures of tau filaments from patients with Alzheimer's disease, which contain both 3R and 4R tau. Here we determine the structures of tau filaments from patients with Pick's disease, a neurodegenerative disorder characterized by frontotemporal dementia. The filaments consist of residues Lys254–Phe378 of 3R tau, which are folded differently from the tau filaments in Alzheimer's disease, establishing the existence of conformers of assembled tau. The observed tau fold in the filaments of patients with Pick's disease explains the selective incorporation of 3R tau in Pick bodies, and the differences in phosphorylation relative to the tau filaments of Alzheimer's disease. Our findings show how tau can adopt distinct folds in the human brain in different diseases, an essential step for understanding the formation and propagation of molecular conformers.
© 2018 Medical Xpress