Calcifications in the eye increase risk for progression to advanced AMD by more than six times

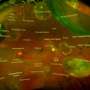
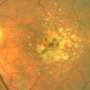
Calcified nodules in the retina are associated with progression to late stages of age-related macular degeneration (AMD). Experts from Queen's University Belfast, working in partnership with the University of Alabama of Birmingham and in collaboration with UK material scientists and US clinical ophthalmology practices, made the ground-breaking discovery that the calcified nodules in the retina – the thin layer of tissue that lines the back of the eye – increase the risk for progression to advanced AMD more than six times.
The findings could revolutionise treatment options for people with age-related macular degeneration (AMD), which is the leading cause of vision impairment in older individuals worldwide.
There is no current treatment for the majority of patients with AMD and irreversible visual loss has been associated with depression and other health problems.
Experts carried out the research as there is an urgent need to identify early events that might lead to visual loss so that these events can be targeted.
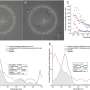
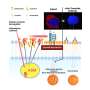
The research, published in the journal Science Translational Medicine, used clinical imaging in patients from ophthalmologists K. Bailey Freund (New York) and Srinivas R. Sadda (Los Angeles), and molecular analysis of eye samples from the laboratory of Dr. Christine Curcio at University of Alabama at Birmingham (UAB), by co-first author Matthew Pilgrim.
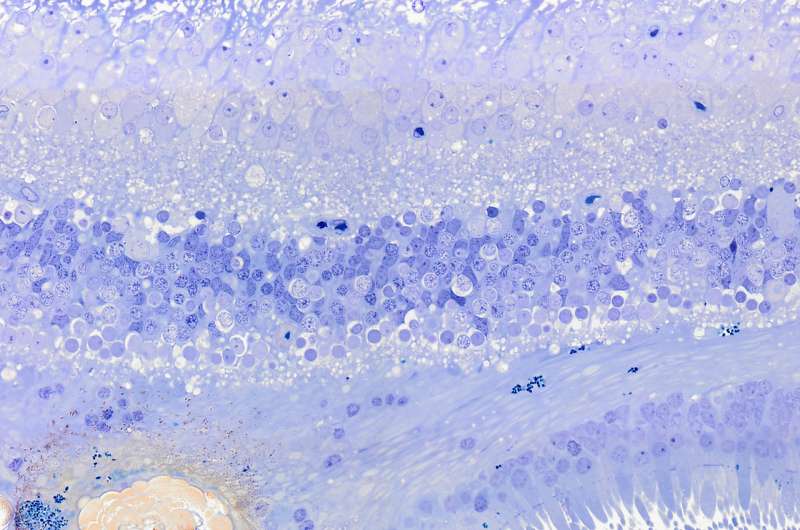
The team discovered that large calcified nodules in the retina were associated with progression of late stages of AMD, especially with the more insidious atrophic form of the disease, of which there are currently no treatment options. The experts believe that with further research and with early intervention, some patients could actually be treated with simple measures such as modifying their diet.
Dr. Imre Lengyel, Senior Lecturer and Researcher in the School of Medicine Dentistry and Biomedical Sciences at Queen's University Belfast, said: "Our research revealed that early changes in the back of the eye can lead to the build-up of hard mineral deposits, made of calcium and phosphate that may incorporate other types of trace metals, like magnesium. The build-up of these mineral deposits are an indicator of irreversible damage of the retina."
Dr. Curcio, Professor in Ophthalmology at University of Alabama, said: "By fully understanding the causes behind the changing environment in which these large, damaging nodules grow we could design new ways to intervene with their growth earlier in the disease process than is currently possible.
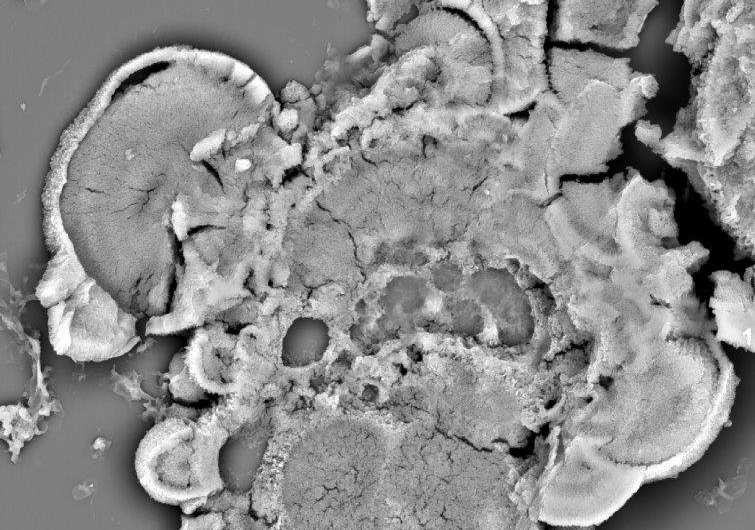
"Identification of these risks associated with disease progression in the eye, especially in the retina, could become a diagnostic tool for monitoring the progression of retinal degeneration. This allows ophthalmologists to counsel their patients more wisely and also allow us to think about slowing or halting the progression of disease, earlier in its course."
Co-first author Anna CS Tan MD said: "Treatment of AMD is expensive, yet so are caregiving or institutional living. Based on the global burden of AMD the cost is estimated to be over 300 billion worldwide." Other co-first author Mathew G Pilgrim continues: "Therefore, delaying progression of AMD, or better yet, minimizing or eliminating risk factors, could lead to a substantial reduction in costs borne by health care payers, individuals, and society.'
Dr. Jayakrishna Ambati, DuPont Guerry, III, Professor of Ophthalmology and Director of the Center for Advanced Vision Science at the University of Virginia, said: "This work will substantially advance the field and spur new thinking in AMD."
The next steps for the research team will be to further their understanding of the disease processes associated with this degeneration. The team will aim to help determine new treatment options for patients, which could be as simple as modification of diet. The research will also allow ophthalmologists to advise their patients about prognosis more fully, based on the details found in their clinical imaging.
More information: Anna C. S. Tan et al. Calcified nodules in retinal drusen are associated with disease progression in age-related macular degeneration, Science Translational Medicine (2018). DOI: 10.1126/scitranslmed.aat4544