November 12, 2018 report
Eyepatch with dissolvable needles used to treat eye disease
A team of researchers affiliated with several institutions in Singapore has developed an eyepatch with dissolvable needles for use in treating eye diseases. In their paper published in the journal Nature Communications, the group describes their eyepatch and how well it worked in mice.
Current methods used to treat eye diseases such as glaucoma and macular degeneration include applying eye drops or using needles to inject drugs. But eyedrops fail to deliver enough drugs and using needles to poke through the cornea can be risky. And neither method allows for applying doses of drugs over an extended period of time. For these reasons, scientists seek a better treatment. In this new effort, the researchers have come up with a new method that does not involve eyedrops or traditional needles—instead, dissolvable needles deliver an initial strong dose and then slowly deliver a secondary dose.
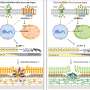
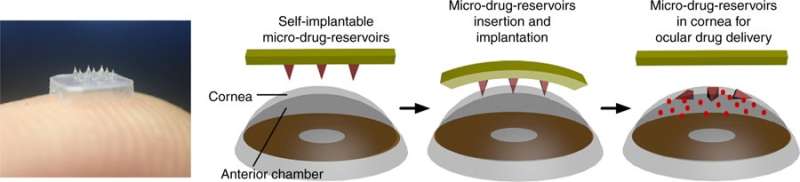
The new delivery method is applied to the front of the eye like a patch on the skin—it is put in place by hand. The eye patch is made in two layers—a base and a span of microneedles. When the patch is applied to the eye, eye fluid seeps into the space between the base and the microneedles, dissolving the material holding them together. At this point, the patch can be removed—the microneedles stay behind, embedded in the cornea. The microneedles are made in two layers, as well. The outer layer dissolves right away, delivering an initial dose of a drug. The inner layer takes several days to dissolve, delivering a secondary dose over time.
The researchers tested their new eye patch on lab mice—each had a case of corneal neovascularization, in which blood vessels grow into the cornea, obscuring vision. Prior research had shown that an antibody called DC101 blocks a protein needed for blood vessel progression. Using the antibody as an ingredient in the microneedles worked as hoped. The researchers report that they were 90 percent effective in treating the disease. More testing is required to determine if there are undesirable side effects before testing can begin in humans.
-

(L-R) Assistant Professor Wang Xiaomeng from NTU's Lee Kong Chian School of Medicine and Professor Chen Peng from the NTU School of Chemical and Biomedical Engineering are part of an NTU team that developed a 'contact lens' eye patch to treat eye diseases. Painless and minimally invasive, the patch could potentially be a viable alternative to the current treatment options, which face poor patient compliance. Credit: NTU Singapore -

The microneedles on the eye patch can be loaded with drugs. Worn like contact lenses, the patch is painless and minimally invasive. The drug is released slowly as the biodegradable microneedles dissolve in the corneal tissue. Credit: NTU Singapore
More information: Aung Than et al. Self-implantable double-layered micro-drug-reservoirs for efficient and controlled ocular drug delivery, Nature Communications (2018). DOI: 10.1038/s41467-018-06981-w
© 2018 Science X Network