New technique for minimally invasive lung cancer surgery—ultrasonic device safe, effective
According to a new study, an ultrasonic vessel-sealing device can improve patient outcomes by reducing the incidence of thoracotomy conversion for VATS/robotic anatomical lung resection. The trial, which included patients in the United States, United Kingdom, and Canada, is the first to evaluate the use of ultrasonic sealing for lobectomy, the most common thoracic surgical procedure.
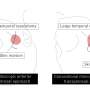
Technology device trials in thoracic surgery are uncommon, with this study being the first intraoperative device study in more than 20 years. 150 patients planned for VATS/robotic anatomical lung resection in seven centers were enrolled. PA Branches of 7mm or less were sealed and divided with an ultrasonic energy vessel-sealing device. The remainder of the lobectomy was performed according to surgeon preference. A total of 424 PA branches were divided; 239 with the ultrasonic vessel-sealing device, 181 with endostaplers, and 4 with endoscopic clips.
Intraoperative, in-hospital, and 30-day post-operative bleeding and complications were observed. 1.3 percent of the PA branches divided with the ultrasonic vessel-sealing device and 2.2 percent of PA branches divided with endostaplers bled intraoperatively. Among the patients with seal failures, one patient required conversion to thoracotomy for vascular repair in the ultrasonic energy group. There was no postoperative bleeding from divided PA branches with either sealing method. One patient was re-operated for hemothorax from a bleeding bronchial artery. Mean and median length of stay was 4.1 and 3.8 days, respectively. There was no mortality at 30-days.
Preliminary results appear to show that use of an ultrasonic sealing device can decrease the rate of conversion to thoracotomy, resulting in safer and less-invasive procedures.
Lead author, Moishe Liberman, MD, Ph.D., of the Division of Thoracic Surgery, University of Montréal, explains, "This is an exciting new application of technology to enable minimally invasive lung surgery for the benefit of patients today and in the future. By promoting less invasive procedures, we promote quicker healing and reduce risks, resulting in improved outcomes for patients."
More information: Prospective, Multi-Center, International Phase 2 Trial Evaluating Ultrasonic Energy for Pulmonary Artery Branch Sealing in VATS Lobectomy. Presented by Moishe Liberman, MD, PhD, Saturday, May 4, 2019 at the AATS 99th Annual Meeting.