Rapid tissue donation program offers feasible approach to improve research
Precision medicine with targeted therapies has led to improved treatment options and patient outcomes. These approaches were developed by studying tumors grown in laboratories and patient samples obtained before and during their treatment. However, there is often a limited supply of patient samples to adequately study, and the samples that exist do not always tell the complete genetic story of how the patient responded to specific drugs and the reasons why they failed treatment. Researchers need a better way to determine how tumors respond to therapies and evolve to resist drug treatment.
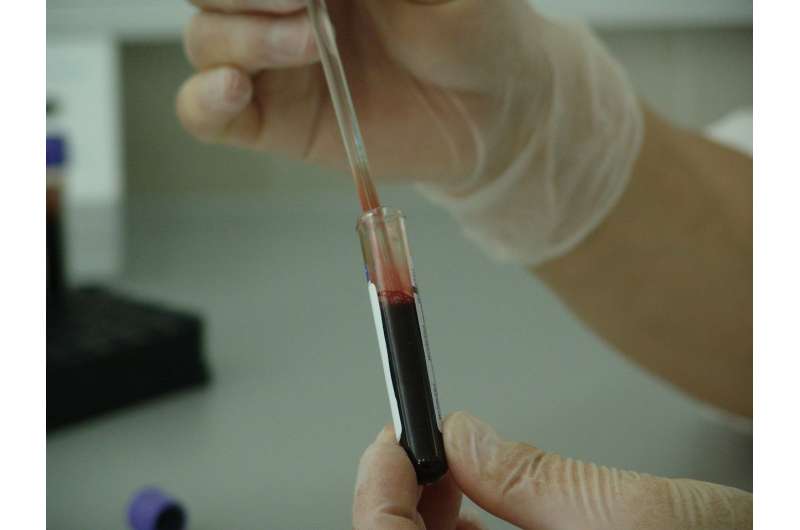
In an article published in Cancer Medicine, Moffitt Cancer Center scientists describe a community-based program called the Rapid Tissue Donation (RTD) protocol. It enables patients to consent to donating tumor tissue and blood samples for research purposes after their death. The samples provided by patients postmortem enable researchers to study the genetic and molecular makeup of primary and metastatic tumors after the patient failed treatment, and to compare those finding with what was known about the patient during earlier phases of their therapy.
There were several challenges that the Moffitt team had to overcome before embarking on this program, including ethical considerations and logistical challenges, communication with hospice care facilities, locating autopsy facilities in the community and identifying tumors in postmortem specimens.
During a two-year span from Nov. 2015 to Nov. 2017, Moffitt researchers were able to gain consent from 21 lung cancer patients from Hillsborough, Pinellas and Pasco counties to participate in the RTD study. They collected 180 tumor tissue and blood specimens from nine deceased patients, while the remaining 12 patients were still alive at the time of the article publication.
One of the logistical challenges the researchers faced was the need to preserve the specimens as quickly as possible after death to ensure that the tissue and molecular material remained intact. The average time to collect the specimens for all nine patients was 15.8 hours. Samples were collected within 20 hours of death for eight donors, and by 41 hours from death for one donor due to unavoidable logistic complexities.
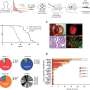
Analysis of the specimens found that most of the DNA samples taken from the primary and metastatic tumors of the same patient were similar and contained the same DNA mutations. The researchers also discovered an AGK-BRAF fusion in one patient with a known EML4-ALK fusion and resistance to ALK inhibitor therapy. Activated BRAF can promote tumor growth. "This is a compelling finding because, had the AGK-BRAF fusion been detected during treatment, physicians could have adapted therapy to include a BRAF-targeted agent," said Eric Haura, M.D., medical oncologist and director of Moffitt's Lung Cancer Center of Excellence.
Analysis of tumor tissue samples for protein biomarkers showed that expression of the protein PD-L1 varied up to 55% among samples taken from the primary tumor and metastatic tumors from the same patient. Levels of PD-L1 are used as a diagnostic assay for lung cancer patients to determine if they should receive certain drugs.
"Twenty to sixty percent of individuals in this study would have a different PD-L1 result if different tumor sites were tested," said Haura. "This illustrates the importance of interpreting PD-L1 results with caution because a large number of patients might not be eligible for immunotherapy based on testing of one tumor site but would be eligible based on testing of a tumor in a different location."
Haura is pleased with the progress the RTD program has made and is excited for what is to come. He hopes that it can be expanded into other tumor types and even developed in partnership with other institutions.
More information: Theresa A. Boyle et al, A community‐based lung cancer rapid tissue donation protocol provides high‐quality drug‐resistant specimens for proteogenomic analyses, Cancer Medicine (2019). DOI: 10.1002/cam4.2670