Novel radiotracer meets gold standard for imaging prostate cancer
The novel radiopharmaceutical 18F-PSMA-1007 is both effective and readily available for detecting malignant prostate cancer lesions, according to research published in the April issue of The Journal of Nuclear Medicine. With this new option and 68Ga-PSMA-11, which is already widely used, nuclear medicine departments will have two effective options for staging of prostate cancer, potentially increasing availability for patients worldwide.
68Ga-PSMA is the most commonly used PSMA-labeled tracer in clinical practice for evaluating extent of disease in prostate cancer patients. However, it is produced by a specific generator and has a short half-life of 68 minutes. "The newly introduced 18F-PSMA-1007 has several advantages over the established 68Ga-PSMA," stated Einat Even-Sapir, MD, Ph.D., director of The Institute for Nuclear Medicine at Tel Aviv Sourasky Medical Center in Tel Aviv, Israel. "These include a longer half-life, favorable pharmacokinetics, central mass production and potentially better spatial resolution."
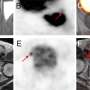
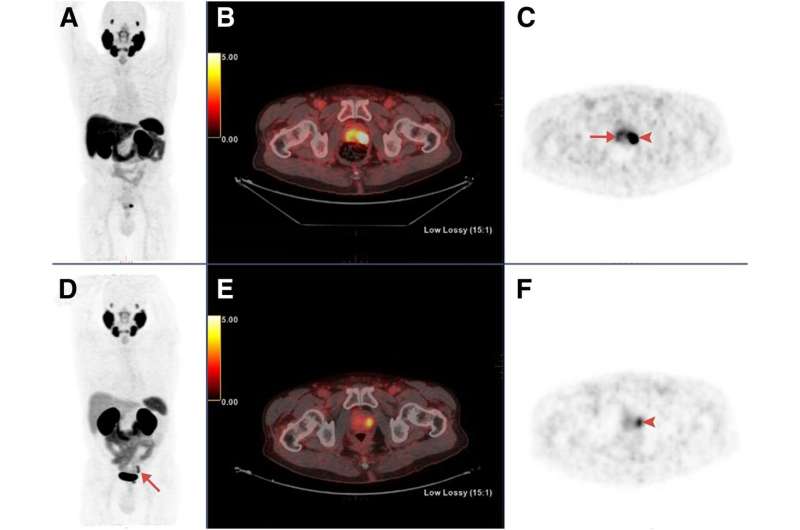
The prospective study aimed to compare the diagnostic accuracy of 18F-PSMA-1007 with 68Ga-PSMA-11 positron emission tomography (PET)/computed tomography (CT) in the same patients presenting with newly diagnosed intermediate- or high-risk prostate cancer. 18F-PSMA-1007 and 68Ga-PSMA-11 PET/CT were performed within 15 days of each other in sixteen patients with intermediate- or high-risk prostate cancer who were scheduled to undergo a radical prostatectomy. Findings from the two PET tracers were compared with histopathologic findings obtained from radical prostatectomy specimens, considered the gold standard.
Labeled PSMA-avid lesions in the prostate were identified in all 16 patients with almost perfect agreement between the two tracers regarding tumor location. Additionally, in four patients, a second positive focus, though less intense, was detected only by 18F-PSMA-1007. Three of these secondary foci were confirmed as areas of prostate cancer, while the fourth was shown on pathological examination to represent chronic prostatitis.
"In view of the near-equal performance of the two tracers, this preliminary study suggests the routine use of 18F-PSMA-1007 in lieu of 68Ga-PSMA-11 for staging prostate cancer patients; clinicians can use either radiotracer based on availability," noted Jonathan Kuten, MD, MHA, nuclear medicine specialist at Tel Aviv Sourasky Medical Center in Tel Aviv, Israel. "It also adds to the collective growing database of evidence supporting the use of PSMA agents for staging intermediate and high-risk patients. We encourage researchers, in the future, to corroborate this study's results in larger cohorts."
More information: Jonathan Kuten et al, Head-to-Head Comparison of 68Ga-PSMA-11 with 18F-PSMA-1007 PET/CT in Staging Prostate Cancer Using Histopathology and Immunohistochemical Analysis as a Reference Standard, Journal of Nuclear Medicine (2019). DOI: 10.2967/jnumed.119.234187