Study demonstrates feasibility of hologram technology in liver tumor ablation
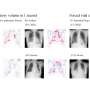
Data from one of the first clinical uses of augmented reality guidance with electromagnetically tracked tools shows that the technology may help doctors quickly, safely, and accurately deliver targeted liver cancer treatments, according to a research abstract presented during a virtual session of the Society of Interventional Radiology's 2020 Annual Scientific Meeting on June 14. The technology provides a three-dimensional holographic view inside a patient's body, allowing interventional radiologists to accurately burn away tumors while navigating to avoid organs and other critical structures.
"Converting traditional two-dimensional imaging into three-dimensional holograms which we can then utilize for guidance using augmented reality helps us to better view a patient's internal structures as we navigate our way to the point of treatment," said Gaurav Gadodia, MD, lead author of the study and radiology resident at Cleveland Clinic. "While conventional imaging like ultrasound and CT is safe, effective, and remains the gold-standard of care, augmented reality potentially improves the visualization of the tumor and surrounding structures, increasing the speed of localization and improving the treating-physician's confidence."
In this initial in-human pilot study, the technology was used to deliver a treatment known as percutaneous thermal ablation of solid liver tumors. To apply this technology, the physicians use multi-phase CT to record coordinate markers placed on a patient's body. This imaging data is added to a software application that allows for segmentation of the tumor and nearby structures within the marked coordinate space. This information is fed into a proprietary augmented reality application, which utilizes Microsoft's HoloLens technology, a virtual reality headset with transparent lenses, to project a segmented hologram of the patient's imaged anatomy directly onto the patient. The hologram is registered to the coordinate markers to ensure accurate location of the relevant anatomy.
Utilizing electromagnetic tracking, instruments including the ablation probe can also be visualized in the augmented reality space during the procedure, thus allowing for true holographic intraprocedural guidance. Interventional radiologists can then use the combination of the holographic images of the patient's anatomy and tracked tools to find the tumor in the patient's liver quickly, check for optimal targeting of the tumor by the ablation probe, and avoid key structures.
The study included five patients who were selected for microwave ablation of their liver tumors. For safety during this IRB-approved study, the gold standard of ultrasound was used for primary clinical decision making and probe guidance, with direct comparison to holographic guidance. Following ablation, images and video from post-procedural sonography, cone beam and multi-detector row CT, and HoloLens recordings were evaluated. In all five cases, intra-procedural holographic guidance was in agreement with the standard ultrasound-based guidance. Post-procedural imaging showed adequate tumor ablation, and no patients experienced tumor recurrence at the three-month follow-up. In this early phase pilot study, the authors anecdotally observed that the speed of tumor localization was faster with holographic guidance, and that their confidence in optimal ablation and critical structure avoidance was improved over standard imaging guidance. They are further attempting to quantify these findings as they continue to enroll patients in the study.
Beyond its use during treatments, interventional radiologists also see value in using this tool for clinicians' planning purposes and for improving patient engagement and understanding of the condition and treatment.
"This technique can be used intra-procedurally to check the accuracy and quality of the treatment, as well as pre-procedurally to engage with the patient in their own care," said Charles Martin, III, MD, FSIR, an interventional radiologist at Cleveland Clinic who is the principal investigator of the IRB and the senior author of the study. "We can change 2-D images into holograms of a patient's distinct anatomy so that both the physician and the patient get a better understanding of the tumor and treatment."
Researchers continue to test this technology for ablations in the abdominal area with plans to expand to other types of procedures and in other areas of the body. The technology has only been tested for feasibility and therefore cannot yet be used as a standalone method for delivering a treatment.
Additional information about the clinical trial is available at ClinicalTrials.gov, using the identifier NCT03500757. This work was funded through internal enterprise grants from Cleveland Clinic, as well as the Ohio-based biotechnology startup, MediView.
More information: Abstract: True 3-D holographic visualization for performance of percutaneous thermal ablation of solid liver tumors (3-D-HPA): An update on in-human evaluation. G. Gadodia; J. Yanof; K. West; S. Al-Nimer; A. Hanlon; C. Weunski; C Martin; Cleveland Clinic Foundation, Lerner Research Institute, Cleveland Clinic, John Carroll University, Cleveland, OH.