Alcohol, nicotine mix during pregnancy increases health risk in newborns

University of Houston researchers have found that during early pregnancy, the mix of alcohol and nicotine significantly alters the gene regulatory pathways of the developing fetus, which can lead to major deficiencies in brain development. Metin Akay, founding chair and John S. Dunn Endowed Chair Professor of biomedical engineering is reporting the findings, the first study of its kind, in the Nature journal Scientific Reports.
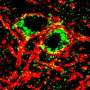
"The alterations of these pathways are crucial since they are involved in neural network formation, cell development and communication," reports Akay. "Among pathways in which many genes and miRNAs were significantly altered in response to perinatal nicotine/alcohol co-exposure are dopamine cell growth, neuronal migration, neuronal axon guidance, neurotrophin signaling and glutamatergic synapse."
Addictive substances act on the brain's reward system by triggering the release of the dopamine hormone through the activation of the mesocorticolimbic DA system, also known as the reward circuitry in the brain.
"A characteristic structure of dopamine neurons are the long axons that project to different regions of the brain to build functional networks, which results in pathways such as the mesocorticolimbic DA system," said Akay. "It is highly likely that axon guidance is modulated in the newborn after perinatal substance abuse and may cause faulty assembly of the network."
The alterations in this pathway cause interruptions in cellular communication and development, and finally, lead to synaptic rearrangements in the plasticity and neurological disorders.
It's no small problem.
Maternal substance abuse (drinking and smoking) during pregnancy increases health risks, including cognitive impairments, lower academic achievement, attention deficit hyperactivity disorder (ADHD), the likelihood of substance abuse in newborns, and may even lead sudden infant death syndrome (SIDS). Despite these harmful effects, more than 10% of pregnant women drink and smoke, according to the Centers for Disease Control.
Following alcohol treatment, 1,257 unique genes were found to be differentially upregulated and 330 were differentially downregulated. Following perinatal nicotine-alcohol treatment contrasted against the alcohol group, 2,113 genes were upregulated and 1,836 were downregulated.
"A more comprehensive treatment needs to be developed for the perinatal co-exposure since more pathways and gene expressions were significantly altered, suggesting the involvement of several addiction pathways in newborns," said Akay.
"Until now, the influence of maternal alcohol and nicotine co-exposure on the brain development of newborns has not been investigated at the multi scale from molecular, to cellular and to systemic levels," said Yasemin Akay, instructional associate professor of biomedical engineering and the co-lead investigator on the project. "Our group has focused on the integration of molecular, cellular and systemic data—using a custom-made implantable dopamine probe and artificial intelligence—to better understand the addiction mechanism and develop effective therapeutics," she said.
More information: Tina Kazemi et al, Investigating the influence of perinatal nicotine and alcohol exposure on the genetic profiles of dopaminergic neurons in the VTA using miRNA–mRNA analysis, Scientific Reports (2020). DOI: 10.1038/s41598-020-71875-1