A first-in-class antibody designed to prevent clotting
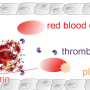
Researchers from Monash University have designed a novel antibody that inhibits one particular blood-borne protein to prevent clot formation, or thrombosis, without potential adverse side effects.
The study, led by Dr. Erik Westein and Associate Professor Christoph Hagemeyer from the Australian Center for Blood Diseases at Monash University, describes how the novel antibody has been engineered to only detect and block the pathological form of the Von Willebrand Factor (VWF) blood protein.
The antibody is able to stop pathological thrombosis that can cause heart attacks and strokes without impacting normal healthy clotting.
The findings have been published in Haematologica.
Heart attack and stroke remain the leading causes of mortality and morbidity worldwide. Current anti-thrombotic (anti-clotting) therapies can, and do, cause severe bleeding complications because they also interfere with normal blood clotting. Four out of five patients who receive anti-platelet therapy still have recurring cardiovascular events.
Existing anti-platelet drugs therefore cannot be used in higher doses. As a result, their efficacy remains disappointingly low and future therapies require a fundamental re-design from the ground up.
"Our approach was to first identify the biological differences between normal blood clotting and pathological blood clotting, and we found that VWF changes its properties when dangerous blood clots are forming," Dr. Westein said.
"Next, we engineered an antibody that only detects and blocks this pathological form of VWF and is therefore only active when a blood clot becomes pathological."
Associate Professor Hagemeyer said: "We analyzed the properties of existing antibodies against VWF and identified optimal properties of each that would bind and block VWF under pathological blood clotting conditions. We then combined these optimal molecular structures into a new antibody to generate a first-in-class drug candidate that has the potential to stop dangerous blood clots without any adverse effects such as bleeding complications."
Dr. Westein said clinicians presently face a delicate balance of drug efficacy versus bleeding side effects. "Our engineered antibody is purposely designed to not interfere with normal blood clotting so we expect that it can be used at a much higher and effective dose compared to existing therapies," he said.
Associate Professor Hagemeyer said the in vitro study worked with human blood samples.
"Our next step will be to test the efficiency of our antibody in small animal models to understand how it works in a complex living system analogous to our own."
More information: Thomas Hoefer et al. Targeting shear gradient activated von Willebrand factor by the novel single-chain antibody A1 reduces occlusive thrombus formation in vitro, Haematologica (2020). DOI: 10.3324/haematol.2020.250761