How zika virus degrades an essential protein for neurological development, getting it to 'eat itself'
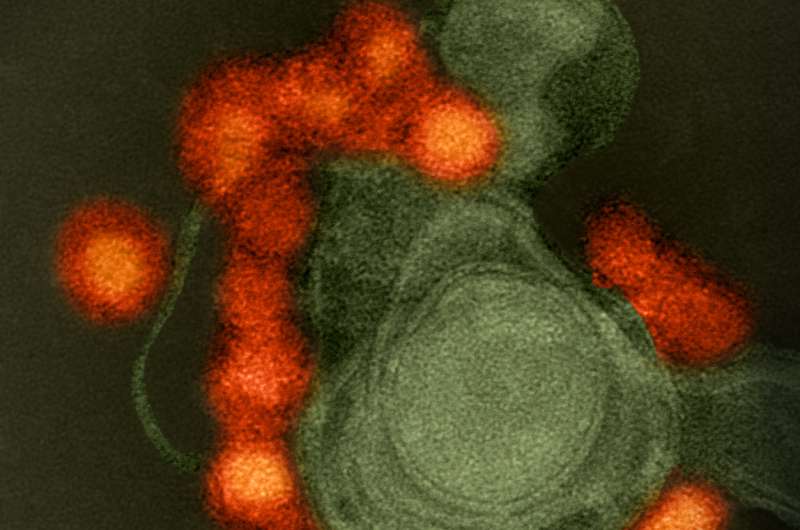
In a study published in Autophagy, researchers at the University of Maryland (UMD) shed new light on how Zika virus hijacks our own cellular machinery to break down a protein that is essential for neurological development and cellular communication, getting it to "eat itself". By triggering this process known as autophagy, Zika virus is able to degrade an important protein, a process that may contribute to the development of neurological or brain deficiencies and congenital birth defects in the newborns of infected pregnant women. By understanding the underlying mechanisms of how this process takes place, researchers are coming closer to developing therapeutic interventions to prevent congenital birth defects such as microcephaly caused by Zika virus infection in pregnant mothers.
"The Zika virus is able to disrupt our cellular mechanisms to create a conducive environment to replicate," explains Yanjin Zhang, associate professor in Veterinary Medicine at UMD. "It upregulates some proteins and downregulates others that have antiviral roles, manipulating and interfering with cells to its own advantage. In this case, it looks like the KPNA2 protein may have some antiviral effects, so the virus uses the natural cellular self-destruction process called autophagy, or self-eating, to get rid of KPNA2."
The human body is full of mechanisms to move information and equipment like proteins around, or clear out unnecessary equipment through self-destruct processes like autophagy. In autophagy, cellular components like proteins are marked as damaged, essentially triggering the cell to eat itself to recycle vital ingredients for cell regeneration. However, in this case, the Zika virus wants the protein KPNA2 destroyed for its own benefit.
"Researchers knew that KPNA2 has important roles in transporting proteins, but we didn't know the mechanism of its turnover until now," says Zhang. "KPNA2 is known to transport important cellular factors needed for development, growth, and cell differentiation during neurological or brain development."
Since Zika virus is known to cause infection in pregnant women that can lead to brain deficiencies and congenital defects like microcephaly (a birth defect leading to smaller head and brain size) in newborns, the fact that it degrades this particular protein is important to understanding the development of these defects.
Zika virus is a globally mosquito-transmitted virus that has been identified in 87 countries and territories as of 2019 according to the World Health Organization (WHO). While no cases have been reported in the United States in the last few years, the WHO reports a suspected total of over 30,000 cases in the Americas alone in 2018. After the Zika virus epidemic in 2015-2016, the health risk for pregnant mothers and congenital birth defects in newborns became a major public health concern.
"Zika virus is more of an issue internationally than locally right now," says Zhang, "but as we can see with COVID-19, a global disease can easily become a local concern. Nowadays, it is so easy to travel from one continent to another, and with commercial trade, everything is interlinked. Vectors like mosquitoes can be carried across the world and open new pathways of transmission."
The changing climate also plays a role in the spread of infectious diseases like this, explains Zhang. "This is vector-borne or mosquito-borne disease, and as the climate changes, the mosquitoes can reproduce more, and can move farther north and into other areas they may not have been before."
Zhang is hopeful, however, that his work will help lead to therapeutic options that can help prevent birth defects caused by Zika virus infection. "Understanding this mechanism is an important step towards understanding how to control the effects of Zika virus, including congenital birth defects," says Zhang.
More information: Jia He et al, Zika virus NS2A protein induces the degradation of KPNA2 (karyopherin subunit alpha 2) via chaperone-mediated autophagy, Autophagy (2020). DOI: 10.1080/15548627.2020.1823122