Unravelling the multifaceted mechanisms of cancer cell death by mutant p53 targeting compound APR-246
Sophia Ceder and Klas Wiman and their colleagues at Department of Oncology-Pathology have together with researchers from Peter MacCallum Cancer Center, University of Melbourne, University of Cambridge and Aprea Therapeutics published a study in EMBO Molecular Medicine that provides novel understanding on the mechanisms of mutant p53 targeting compound APR-246.
Around half of all tumors carry mutations in the tumor suppressor gene TP53 which is often associated with poor prognosis. The molecule APR-246 (Eprenetapopt) was discovered by Wiman and colleagues around 20 years ago (Bykov et al 2002) and is today the clinically most advanced compound that targets mutant p53. Currently, APR-246 is undergoing a Phase III clinical trial in mutant TP53 myelodysplastic syndrome (MDS) and several Phase II studies in various indications. Earlier this year APR-246 received FDA Breakthrough Therapy Designation for the combination with Azacitidine for the treatment of mutant TP53 MDS and recently also received FDA Fast Track Designation in mutant TP53 acute myeloid leukemia (AML).
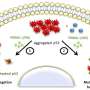
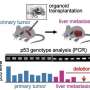
The study investigates the multifaceted mechanisms of action of APR-246 to improve therapeutic efficacy and increase our understanding of APR-246 pharmacodynamics in cancer cells. APR-246 targets mutant p53 and also induces oxidative stress by binding glutathione (GSH) or inhibiting antioxidant enzymes, both major mechanisms of APR-246-mediated cancer cell death. MQ, the active conversion product of APR‐246, forms reversible conjugates with the major antioxidant GSH. The study shows that this complex (GSH bound-MQ) can be exported through the multidrug resistance‐associated protein 1 (MRP1) efflux pump and that blocking MRP1 traps MQ inside cancer cells. This forms an intracellular drug reservoir that may enhance targeting of mutant p53 and disrupt the intracellular redox balance. Blocking MRP1 in combination with APR-246 expands the therapeutic window and leads to pronounced synergistic cancer cell death in vitro in cancer cell lines, ex vivo in patient-derived organoids and in vivo in mice with tumor xenografts.
In conclusion, the study explains the dynamics of APR‐246 and MQ in cancer cells and indicates that MRP1 can play a key role in the sensitivity to APR‐246. Altogether our findings suggest that combination treatment with APR‐246 and drugs that target the redox balance may allow more efficient cancer therapy.
More information: Sophia Ceder et al. A thiol‐bound drug reservoir enhances APR‐246‐induced mutant p53 tumor cell death, EMBO Molecular Medicine (2020). DOI: 10.15252/emmm.201910852