Experimental drug for Alzheimer's disease shows promise
Drug maker Eli Lilly on Monday announced promising results from a small study of an experimental Alzheimer's disease drug called donanemab.
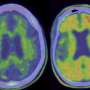
The two-year, phase 2 clinical trial included 272 patients with mild-to-moderate Alzheimer's disease symptoms. The company said that patients who received the drug by infusion every four weeks had a 32 percent slower rate of mental decline than those who were given a placebo, The New York Times reported. After six to 12 months of treatment with the drug, patients no longer had amyloid protein plaques that are hallmarks of Alzheimer disease, according to Daniel Skovronsky, M.D., the company's chief scientific officer.
However, the findings have not been reviewed by other researchers and have not been published in any form, The Times reported. The main side effect was one often seen in patients who take experimental monoclonal antibodies to treat Alzheimer's disease: an accumulation of fluid in the brain. It occurred in close to 30 percent of patients, Skovronsky said, but most had no symptoms. The effect was seen on brain imaging.
The trial findings need to be replicated, Michael Weiner, M.D., a leading Alzheimer's disease researcher at the University of California, San Francisco, told The Times. Even so, "this is big news" and "holds out hope for patients and their families," Weiner added.
More information: The New York Times Article
Copyright © 2020 HealthDay. All rights reserved.